The Effect of an Irradiation-Induced Recombination Suppressing Inversion on the Genetic Stability and Biological Quality of a White Eye-Based Aedes aegypti Genetic Sexing Strain
- PMID: 36292893
- PMCID: PMC9604213
- DOI: 10.3390/insects13100946
The Effect of an Irradiation-Induced Recombination Suppressing Inversion on the Genetic Stability and Biological Quality of a White Eye-Based Aedes aegypti Genetic Sexing Strain
Abstract
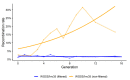
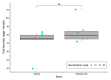
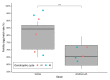
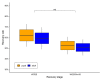
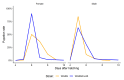
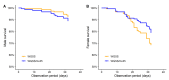
Aedes aegypti is the primary vector of diseases such as dengue, chikungunya, Zika fever, and yellow fever. The sterile insect technique (SIT) has been proposed as a species-specific and environment-friendly tool for the suppression of mosquito vector populations as a major component of integrated vector management strategies. As female mosquitoes are blood-feeders and may transmit pathogenic microorganisms, mosquito SIT depends on the release of sterile males. Genetic sexing strains (GSS) can be used for the efficient and robust separation of males from females. Two Ae. aegypti GSS were recently developed by exploiting eye colour mutations, resulting in the Red-eye GSS (RGSS) and the White-eye GSS (WGSS). In this study, we compared two WGSS, with and without the chromosomal inversion 35 (Inv35), and evaluated their biological quality, including genetic stability. Our results suggest that the WGSS/Inv35 presents a low recombination rate and long-term genetic stability when recombinants are removed from the colony (filtering) and a slow accumulation of recombinants when they are not removed from the colony (non-filtering). The two strains were similar with respect to fecundity, pupal and adult recovery rates, pupation curve, and pupal weight. However, differences were detected in fertility, survival rate of females, and flight ability of males. The WGSS/Inv35 presented lower fertility, higher survival rate of females, and better flight ability of males compared to the WGSS.
Keywords: Zika; chromosomal inversion; dengue; sterile insect technique; vector control.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Genetic Stability and Fitness of Aedes aegypti Red-Eye Genetic Sexing Strains With Pakistani Genomic Background for Sterile Insect Technique Applications.Front Bioeng Biotechnol. 2022 Mar 31;10:871703. doi: 10.3389/fbioe.2022.871703. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 35433649 Free PMC article.
-
Introgression of the Aedes aegypti Red-Eye Genetic Sexing Strains Into Different Genomic Backgrounds for Sterile Insect Technique Applications.Front Bioeng Biotechnol. 2022 Feb 2;10:821428. doi: 10.3389/fbioe.2022.821428. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 35186905 Free PMC article.
-
Genetic sexing strains for the population suppression of the mosquito vector Aedes aegypti.Philos Trans R Soc Lond B Biol Sci. 2021 Feb 15;376(1818):20190808. doi: 10.1098/rstb.2019.0808. Epub 2020 Dec 28. Philos Trans R Soc Lond B Biol Sci. 2021. PMID: 33357054 Free PMC article.
-
Could species-focused suppression of Aedes aegypti, the yellow fever mosquito, and Aedes albopictus, the tiger mosquito, affect interacting predators? An evidence synthesis from the literature.Pest Manag Sci. 2022 Jul;78(7):2729-2745. doi: 10.1002/ps.6870. Epub 2022 Apr 7. Pest Manag Sci. 2022. PMID: 35294802 Free PMC article. Review.
-
Towards mosquito sterile insect technique programmes: exploring genetic, molecular, mechanical and behavioural methods of sex separation in mosquitoes.Acta Trop. 2014 Apr;132 Suppl:S178-87. doi: 10.1016/j.actatropica.2013.08.015. Epub 2013 Aug 28. Acta Trop. 2014. PMID: 23994521 Review.
References
-
- Colón-González F.J., Sewe M.O., Tompkins A.M., Sjödin H., Casallas A., Rocklöv J., Caminade C., Lowe R. Projecting the Risk of Mosquito-Borne Diseases in a Warmer and More Populated World: A Multi-Model, Multi-Scenario Intercomparison Modelling Study. Lancet Planet. Health. 2021;5:e404–e414. doi: 10.1016/S2542-5196(21)00132-7. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

