Electrochemically Activated CNT Sheet as a Cathode for Zn-CO2 Batteries
- PMID: 36293460
- PMCID: PMC9604323
- DOI: 10.3390/ijms232012602
Electrochemically Activated CNT Sheet as a Cathode for Zn-CO2 Batteries
Abstract
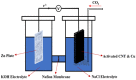
High demand for electrochemical storage devices is increasing the need for high-performance batteries. A Zn-CO2 battery offers a promising solution for CO2 reduction as well as energy storage applications. For this study, a Zn-CO2 battery was fabricated using a Carbon Nanotube (CNT) sheet as a cathode and a Zn plate as an anode. The electrochemical activation technique was used to increase the surface area of the CNT electrode by roughly 4.5 times. Copper (Cu) as a catalyst was then deposited onto the activated CNT electrode using electrodeposition method and different Cu loadings were investigated to optimize CO2 reduction. The final assembled Zn-CO2 battery has a 1.6 V output voltage at a current density of 0.063 mA/cm2, which is higher than most devices reported in the literature. This study demonstrates the importance of activation process which enabled more catalyst loading on the cathode resulted in additional active sites for electroreduction process. This paper presents the activated CNT sheet as a promising cathode material for Zn-CO2 batteries.
Keywords: CNT/Cu composite; CO2 reduction; Zn-CO2 battery.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







Similar articles
-
Effective Ru/CNT Cathode for Rechargeable Solid-State Li-CO2 Batteries.ACS Appl Mater Interfaces. 2021 Sep 22;13(37):44266-44273. doi: 10.1021/acsami.1c11000. Epub 2021 Sep 8. ACS Appl Mater Interfaces. 2021. PMID: 34494812
-
Spinel Zinc Cobalt Oxide (ZnCo2O4) Porous Nanorods as a Cathode Material for Highly Durable Li-CO2 Batteries.ACS Appl Mater Interfaces. 2020 Apr 15;12(15):17353-17363. doi: 10.1021/acsami.9b21347. Epub 2020 Apr 6. ACS Appl Mater Interfaces. 2020. PMID: 32202752
-
High-Performance Zn-I2 Batteries Enabled by a Metal-Free Defect-Rich Carbon Cathode Catalyst.ACS Appl Mater Interfaces. 2023 May 31;15(21):25558-25566. doi: 10.1021/acsami.3c03134. Epub 2023 May 17. ACS Appl Mater Interfaces. 2023. PMID: 37198728
-
Filled Carbon Nanotubes as Anode Materials for Lithium-Ion Batteries.Molecules. 2020 Feb 27;25(5):1064. doi: 10.3390/molecules25051064. Molecules. 2020. PMID: 32120977 Free PMC article. Review.
-
Bifunctional Catalysts for CO2 Reduction and O2 Evolution: A Pivotal for Aqueous Rechargeable Zn-CO2 Batteries.Adv Mater. 2024 Aug;36(35):e2407099. doi: 10.1002/adma.202407099. Epub 2024 Jul 7. Adv Mater. 2024. PMID: 38924576 Review.
Cited by
-
Multifunctional Nanomaterials: Synthesis, Properties, and Applications 2.0.Int J Mol Sci. 2023 Apr 21;24(8):7619. doi: 10.3390/ijms24087619. Int J Mol Sci. 2023. PMID: 37108782 Free PMC article.
References
-
- Qiao Y., Xu S., Liu Y., Dai J., Xie H., Yao Y., Mu X., Chen C., Kline D.J., Hitz E.M., et al. Transient, in situ synthesis of ultrafine ruthenium nanoparticles for a high-rate Li–CO2 battery. Energy Environ. Sci. 2019;12:1100–1107. doi: 10.1039/C8EE03506G. - DOI
-
- Xu S., Das S.K., Archer L.A. The Li-CO2 battery: A novel method for CO2 capture and utilization. RSC Adv. 2013;3:6656–6660. doi: 10.1039/c3ra40394g. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

