Randomized Controlled Trials on Renin Angiotensin Aldosterone System Inhibitors in Chronic Kidney Disease Stages 3-5: Are They Robust? A Fragility Index Analysis
- PMID: 36294504
- PMCID: PMC9605379
- DOI: 10.3390/jcm11206184
Randomized Controlled Trials on Renin Angiotensin Aldosterone System Inhibitors in Chronic Kidney Disease Stages 3-5: Are They Robust? A Fragility Index Analysis
Abstract
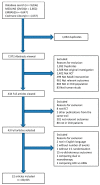
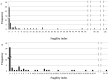
Inhibition of the renin-angiotensin-aldosterone system (RAAS) is broadly recommended in many nephrological guidelines to prevent chronic kidney disease (CKD) progression. This work aimed to analyze the robustness of randomized controlled trials (RCTs) investigating the renal and cardiovascular outcomes in CKD stages 3-5 patients treated with RAAS inhibitors (RAASi). We searched for RCTs in MEDLINE (PubMed), EMBASE databases, and the Cochrane register. Fragility indexes (FIs) for every primary and secondary outcome were calculated according to Walsh et al., who first described this novel metric, suggesting 8 as the cut-off to consider a study robust. Spearman coefficient was calculated to correlate FI to p value and sample size of statistically significant primary and secondary outcomes. Twenty-two studies met the inclusion criteria, including 80,455 patients. Sample size considerably varied among the studies (median: 1693.5, range: 73-17,276). The median follow-up was 38 months (range 24-58). The overall median of both primary and secondary outcomes was 0 (range 0-117 and range 0-55, respectively). The median of FI for primary and secondary outcomes with a p value lower than 0.05 was 6 (range: 1-117) and 7.5 (range: 1-55), respectively. The medians of the FI for primary outcomes with a p value lower than 0.05 in CKD and no CKD patients were 5.5 (range 1-117) and 22 (range 1-80), respectively. Only a few RCTs have been shown to be robust. Our analysis underlined the need for further research with appropriate sample sizes and study design to explore the real potentialities of RAASi in the progression of CKD.
Keywords: chronic kidney disease 1; fragility index; randomized controlled trials; renin-angiotensin-aldosterone system inhibitors; robustness.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Renoprotective RAAS inhibition does not affect the association between worse renal function and higher plasma aldosterone levels.BMC Nephrol. 2017 Dec 20;18(1):370. doi: 10.1186/s12882-017-0789-x. BMC Nephrol. 2017. PMID: 29262813 Free PMC article. Clinical Trial.
-
Management of Renin-Angiotensin-Aldosterone System blockade in patients admitted to hospital with confirmed coronavirus disease (COVID-19) infection (The McGill RAAS-COVID- 19): A structured summary of a study protocol for a randomized controlled trial.Trials. 2021 Feb 5;22(1):115. doi: 10.1186/s13063-021-05080-4. Trials. 2021. PMID: 33546734 Free PMC article.
-
Robustness of Longitudinal Safety and Efficacy After Paclitaxel-Based Endovascular Therapy for Treatment of Femoro-Popliteal Artery Occlusive Disease: An Updated Systematic Review and Meta-Analysis of Randomized Controlled Trials.Ann Vasc Surg. 2024 Apr;101:164-178. doi: 10.1016/j.avsg.2023.11.024. Epub 2023 Dec 26. Ann Vasc Surg. 2024. PMID: 38154491 Free PMC article.
-
The effect of renin-angiotensin-aldosterone system inhibitors on continuous and binary kidney outcomes in subgroups of patients with diabetes: a meta-analysis of randomized clinical trials.BMC Nephrol. 2022 Apr 28;23(1):161. doi: 10.1186/s12882-022-02763-1. BMC Nephrol. 2022. PMID: 35484505 Free PMC article.
-
Discontinuation of Renin-Angiotensin-Aldosterone System Inhibitors Secondary to Hyperkalemia Translates into Higher Cardiorenal Outcomes.Am J Nephrol. 2023;54(7-8):258-267. doi: 10.1159/000531102. Epub 2023 May 18. Am J Nephrol. 2023. PMID: 37231821 Free PMC article.
Cited by
-
Real-world management of chronic and postprandial hyperkalemia in CKD patients treated with patiromer: a single-center retrospective study.J Nephrol. 2024 May;37(4):1077-1084. doi: 10.1007/s40620-024-01897-9. Epub 2024 Feb 6. J Nephrol. 2024. PMID: 38319545
-
Statistical fragility of reporting hemidiaphragmatic paralysis after brachial plexus blocks in randomized controlled trials: a systematic review.Anesthesiol Perioper Sci. 2023;1(2):11. doi: 10.1007/s44254-023-00006-6. Epub 2023 Apr 24. Anesthesiol Perioper Sci. 2023. PMID: 40476887 Free PMC article. Review.
References
-
- Kidney Disease Outcomes Quality Initiative (K/DOQI) K/DOQI clinical practice guidelines on hypertension and antihyper-tensive agents in chronic kidney disease. Am. J. Kidney Dis. 2004;43((Suppl. 1)):S1–S290. - PubMed
-
- Taler S.J., Agarwal R., Bakris G.L., Flynn J.T., Nilsson P.M., Rahman M., Sanders P.W., Textor S.C., Weir M.R., Townsend R.R., et al. KDOQI US commentary on the 2012 KDIGO clinical practice guideline for man-agement of blood pressure in CKD. Am. J. Kidney Dis. 2013;62:201–213. doi: 10.1053/j.ajkd.2013.03.018. - DOI - PMC - PubMed
-
- Xie X., Liu Y., Perkovic V., Li X., Ninomiya T., Hou W., Zhao N., Liu L., Lv J., Zhang H., et al. Renin-Angiotensin system inhibitors and kidney and cardiovascular outcomes in patients with CKD: A Bayesian network meta-analysis of randomised clinical trials. Am. J. Kidney Dis. 2016;67:728–741. doi: 10.1053/j.ajkd.2015.10.011. - DOI - PubMed
-
- Walsh M., Srinathan S.K., McAuley D.F., Mrkobrada M., Levine O., Ribic C., Molnar A.O., Dattani N.D., Burke A., Guyatt G., et al. The statistical significance of randomized controlled trial results is frequently fragile: A case for a Fragility Index. J. Clin. Epidemiol. 2014;67:622–628. doi: 10.1016/j.jclinepi.2013.10.019. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

