The Small Ras Superfamily GTPase Rho4 of the Maize Anthracnose Fungus Colletotrichum graminicola Is Required for β-1,3-glucan Synthesis, Cell Wall Integrity, and Full Virulence
- PMID: 36294561
- PMCID: PMC9604917
- DOI: 10.3390/jof8100997
The Small Ras Superfamily GTPase Rho4 of the Maize Anthracnose Fungus Colletotrichum graminicola Is Required for β-1,3-glucan Synthesis, Cell Wall Integrity, and Full Virulence
Abstract
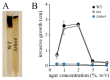
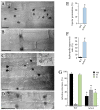
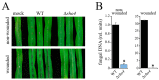
Small Ras superfamily GTPases are highly conserved regulatory factors of fungal cell wall biosynthesis and morphogenesis. Previous experiments have shown that the Rho4-like protein of the maize anthracnose fungus Colletotrichum graminicola, formerly erroneously annotated as a Rho1 protein, physically interacts with the β-1,3-glucan synthase Gls1 (Lange et al., 2014; Curr. Genet. 60:343-350). Here, we show that Rho4 is required for β-1,3-glucan synthesis. Accordingly, Δrho4 strains formed distorted vegetative hyphae with swellings, and exhibited strongly reduced rates of hyphal growth and defects in asexual sporulation. Moreover, on host cuticles, conidia of Δrho4 strains formed long hyphae with hyphopodia, rather than short germ tubes with appressoria. Hyphopodia of Δrho4 strains exhibited penetration defects and often germinated laterally, indicative of cell wall weaknesses. In planta differentiated infection hyphae of Δrho4 strains were fringy, and anthracnose disease symptoms caused by these strains on intact and wounded maize leaf segments were significantly weaker than those caused by the WT strain. A retarded disease symptom development was confirmed by qPCR analyses. Collectively, we identified the Ras GTPase Rho4 as a new virulence factor of C. graminicola.
Keywords: Colletotrichum graminicola; Rho GTPases; Zea mays; appressoria; fungal cell walls; hyphopodia; penetration defect; virulence factor; β-1,3-glucan synthesis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Infection structure-specific expression of β-1,3-glucan synthase is essential for pathogenicity of Colletotrichum graminicola and evasion of β-glucan-triggered immunity in maize.Plant Cell. 2013 Jun;25(6):2356-78. doi: 10.1105/tpc.112.103499. Epub 2013 Jun 28. Plant Cell. 2013. PMID: 23898035 Free PMC article.
-
The galactose metabolism genes UGE1 and UGM1 are novel virulence factors of the maize anthracnose fungus Colletotrichum graminicola.Mol Microbiol. 2024 May;121(5):912-926. doi: 10.1111/mmi.15242. Epub 2024 Feb 23. Mol Microbiol. 2024. PMID: 38400525
-
A chitin synthase with a myosin-like motor domain is essential for hyphal growth, appressorium differentiation, and pathogenicity of the maize anthracnose fungus Colletotrichum graminicola.Mol Plant Microbe Interact. 2007 Dec;20(12):1555-67. doi: 10.1094/MPMI-20-12-1555. Mol Plant Microbe Interact. 2007. PMID: 17990963
-
Fungal Pathogenesis-Related Cell Wall Biogenesis, with Emphasis on the Maize Anthracnose Fungus Colletotrichum graminicola.Plants (Basel). 2022 Mar 23;11(7):849. doi: 10.3390/plants11070849. Plants (Basel). 2022. PMID: 35406829 Free PMC article. Review.
-
Maize Anthracnose Stalk Rot in the Genomic Era.Plant Dis. 2022 Sep;106(9):2281-2298. doi: 10.1094/PDIS-10-21-2147-FE. Epub 2022 Aug 12. Plant Dis. 2022. PMID: 35291814 Review.
References
-
- Cosio E.G., Feger M., Miller C.J., Antelo L., Ebel J. High affinity binding of fungal β-glucan elicitors to cell membranes of species of the plant family Fabaceae. Planta. 1996;200:92–99. doi: 10.1007/BF00196654. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

