The Drug H+ Antiporter FgQdr2 Is Essential for Multiple Drug Resistance, Ion Homeostasis, and Pathogenicity in Fusarium graminearum
- PMID: 36294574
- PMCID: PMC9605015
- DOI: 10.3390/jof8101009
The Drug H+ Antiporter FgQdr2 Is Essential for Multiple Drug Resistance, Ion Homeostasis, and Pathogenicity in Fusarium graminearum
Abstract
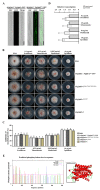
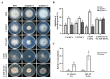
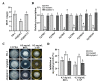
Increased emergence of drug resistance and DON pollution pose a severe problem in Fusarium head blight (FHB) control. While the H+ antiporter (DHA) family plays crucial roles in drug resistance, the characterization of DHA transporters has not been systematically studied in pathogenetic fungi. In this study, a systematic gene deletion analysis of all putative DHA transporter genes was carried out in Fusarium graminearum, and one DHA1 transporter FgQdr2 was found to be involved in multiple drug resistance, ion homeostasis, and virulence. Further exploration showed that FgQdr2 is mainly localized in the cell membrane; its expression under normal growth conditions is comparatively low, but sufficient for the regulation of drug efflux. Additionally, investigation of its physiological substrates demonstrated that FgQdr2 is essential for the transport of K+, Na+, Cu2+, and the regulation of the membrane proton gradient. For its roles in the FHB disease cycle, FgQdr2 is associated with fungal infection via regulating the biosynthesis of virulence factor deoxynivalenol (DON), the scavenging of the phytoalexin, as well as both asexual and sexual reproduction in F. graminearum. Overall, the results of this study reveal the crucial roles of FgQdr2 in multiple drug resistance, ion homeostasis, and pathogenicity, which advance the understanding of the DHA transporters in pathogenetic fungi.
Keywords: Fusarium head blight; H+ antiporter (DHA); multiple drug resistance; virulence.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
The Dynamin-Like GTPase FgSey1 Plays a Critical Role in Fungal Development and Virulence in Fusarium graminearum.Appl Environ Microbiol. 2020 May 19;86(11):e02720-19. doi: 10.1128/AEM.02720-19. Print 2020 May 19. Appl Environ Microbiol. 2020. PMID: 32220839 Free PMC article.
-
Genome-Wide Characterization of PX Domain-Containing Proteins Involved in Membrane Trafficking-Dependent Growth and Pathogenicity of Fusarium graminearum.mBio. 2021 Dec 21;12(6):e0232421. doi: 10.1128/mBio.02324-21. Epub 2021 Dec 21. mBio. 2021. PMID: 34933449 Free PMC article.
-
The Golgin Protein RUD3 Regulates Fusarium graminearum Growth and Virulence.Appl Environ Microbiol. 2021 Feb 26;87(6):e02522-20. doi: 10.1128/AEM.02522-20. Print 2021 Feb 26. Appl Environ Microbiol. 2021. PMID: 33452023 Free PMC article.
-
Recent advances in genes involved in secondary metabolite synthesis, hyphal development, energy metabolism and pathogenicity in Fusarium graminearum (teleomorph Gibberella zeae).Biotechnol Adv. 2014 Mar-Apr;32(2):390-402. doi: 10.1016/j.biotechadv.2013.12.007. Epub 2014 Jan 2. Biotechnol Adv. 2014. PMID: 24389085 Review.
-
Transcriptomics of cereal-Fusarium graminearum interactions: what we have learned so far.Mol Plant Pathol. 2018 Mar;19(3):764-778. doi: 10.1111/mpp.12561. Epub 2017 Jun 7. Mol Plant Pathol. 2018. PMID: 28411402 Free PMC article. Review.
Cited by
-
Osmotically Activated Anion Current of Phycomyces Blakesleeanus-Filamentous Fungi Counterpart to Vertebrate Volume Regulated Anion Current.J Fungi (Basel). 2023 May 31;9(6):637. doi: 10.3390/jof9060637. J Fungi (Basel). 2023. PMID: 37367573 Free PMC article.
-
The Mechanisms of Developing Fungicide Resistance in Fusarium graminearum Causing Fusarium Head Blight and Fungicide Resistance Management.Pathogens. 2024 Nov 18;13(11):1012. doi: 10.3390/pathogens13111012. Pathogens. 2024. PMID: 39599565 Free PMC article. Review.
-
Endophytic strategies decoded by genome and transcriptome analysis of Fusarium nematophilum strain NQ8GII4.Front Microbiol. 2025 Jan 15;15:1487022. doi: 10.3389/fmicb.2024.1487022. eCollection 2024. Front Microbiol. 2025. PMID: 39881987 Free PMC article.
-
The Dual Pathogen Fusarium: Diseases, Incidence, Azole Resistance, and Biofilms.J Fungi (Basel). 2025 Apr 9;11(4):294. doi: 10.3390/jof11040294. J Fungi (Basel). 2025. PMID: 40278115 Free PMC article. Review.
-
The Non-Histone Protein FgNhp6 Is Involved in the Regulation of the Development, DON Biosynthesis, and Virulence of Fusarium graminearum.Pathogens. 2024 Jul 16;13(7):592. doi: 10.3390/pathogens13070592. Pathogens. 2024. PMID: 39057819 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources

