2,4-Diacetylphloroglucinol Modulates Candida albicans Virulence
- PMID: 36294582
- PMCID: PMC9604888
- DOI: 10.3390/jof8101018
2,4-Diacetylphloroglucinol Modulates Candida albicans Virulence
Abstract
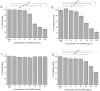
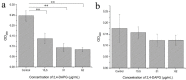
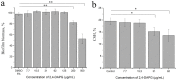
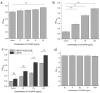
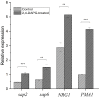
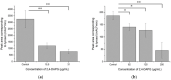
The dimorphic fungus Candida albicans is one of the most important opportunistic pathogens for humankind. The use of fungicides against Candida could be associated with sub-inhibitory effects, which are referred to as fungal stress responses and are undesirable for the host. In this work, we investigated the antifungal action of 2,4-diacetylphloroglucinol (2,4-DAPG) against Candida albicans ATCC 10231 with a focus on their biofilm-forming ability. We found that 2,4-DAPG was able to reduce the ability of Candida cells to form biofilms, but complete inhibition and eradication effects were not achieved. Furthermore, C. albicans cells in the adherent state were characterized by reduced susceptibility to 2,4-DAPG compared to planktonic cells. The investigation of the mechanisms that could explain the antibiofilm action of 2,4-DAPG revealed a reduction in the cell`s surface hydrophobicity and the inhibition of the yeast-to-hyphae transition. The inhibition of the Candida cells filamentation was accompanied by an increase in the expression of the NRG1 gene, which is a negative regulator of hyphal development. In addition, we microscopically visualized the treated biofilms and revealed numerous channels that were decorated with particles and localized on the hyphae. We assumed that these hyphal structures could be associated with the secretion of aspartyl proteases (Sap). The performed assessments revealed an increase in the activity of Sap, which was accompanied by an increase in the expression of the sap2 and sap4 genes. The antifungal action of 2,4-DAPG is known to be associated with affecting the permeability of cellular structures, which leads to H+ATPase malfunction and the disruption of mitochondrial respiration. The subsequent cytosol acidification and generation of ROS trigger the inhibition of Candida filamentation and activation of Sap production. The introduction of antioxidant Trolox simultaneously with 2,4-DAPG leads to a reduction in Sap production. Collectively, the obtained data indicate new aspects of the interaction of fungal cells with 2,4-DAPG, an antimicrobial metabolite of Pseudomonas spp.
Keywords: 2,4-diacetylphloroglucinol; Candida albicans; aspartyl protease; biofilm; hyphal channels; virulence.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Keel C., Weller D.M., Natsch A., Défago G., Cook R.J., Thomashow L.S. Conservation of the 2,4-diacetylphloroglucinol biosynthesis locus among fluorescent Pseudomonas strains from diverse geographic locations. Appl. Environ. Microbiol. 1996;62:552–563. doi: 10.1128/aem.62.2.552-563.1996. - DOI - PMC - PubMed
-
- Troppens D., Moynihan J., Barret M., O’Gara F., Morrissey J. Genetics and Evolution of 2,4-Diacetylphloroglucinol Synthesis in Pseudomonas fluorescens. In: Bruijn F.J., editor. Molecular Microbial Ecology of the Rhizosphere. John Wiley & Sons; Hoboken, NJ, USA: 2013. pp. 593–605.
-
- Kwak Y.S., Han S., Thomashow L.S., Rice J.T., Paulitz T.C., Kim D., Weller D.M. Saccharomyces cerevisiae genome-wide mutant screen for sensitivity to 2,4-diacetylphloroglucinol, an antibiotic produced by Pseudomonas fluorescens. Appl. Environ. Microbiol. 2011;77:1770–1776. doi: 10.1128/AEM.02151-10. - DOI - PMC - PubMed
-
- Troppens D., Dmitriev R., Papkovsky D., O’Gara F., Morrissey J. Genome-wide investigation of cellular targets and mode of action of the antifungal bacterial metabolite 2,4-diacetylphloroglucinol in Saccharomyces cerevisiae. FEMS Yeast Res. 2013;13:322–334. doi: 10.1111/1567-1364.12037. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

