Highlighting Bacteria with Calcifying Abilities Suitable to Improve Mortar Properties
- PMID: 36295324
- PMCID: PMC9612027
- DOI: 10.3390/ma15207259
Highlighting Bacteria with Calcifying Abilities Suitable to Improve Mortar Properties
Abstract
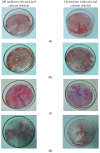
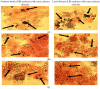
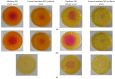
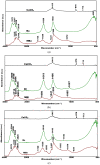
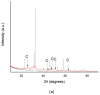
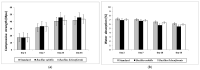
Biomineralization, the use of microorganisms to produce calcium carbonate, became a green solution for application in construction materials to improve their strength and durability. The calcifying abilities of several bacteria were investigated by culturing on a medium with urea and calcium ions. The characterization of the precipitates from bacterial cultures was performed using X-ray diffraction, Fourier transform infrared spectroscopy, and thermogravimetric analysis. The formation of carbonate crystals was demonstrated by optical and scanning electron microscopy. Water absorption and compressive strength measurements were applied to mortars embedded with sporal suspension. The efficiency of the supplementation of mortar mixtures with bacterial cells was evaluated by properties, namely the compressive strength and the water absorption, which are in a relationship of direct dependence, the increase in compressive strength implying the decrease in water absorption. The results showed that Bacillus subtilis was the best-performing bacterium, its introduction into the mortar producing an increase in compressive strength by 11.81% and 9.50%, and a decrease in water absorption by 11.79% and 10.94%, after 28 and 56 days of curing, respectively, as compared to standards. The exploitation of B. subtilis as a calcifying agent can be an interesting prospect in construction materials.
Keywords: Bacillus; biomineralization; concrete; ureolytic bacteria.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Investigation of Self-Healing Mortars with and without Bagasse Ash at Pre- and Post-Crack Times.Materials (Basel). 2022 Feb 23;15(5):1650. doi: 10.3390/ma15051650. Materials (Basel). 2022. PMID: 35268883 Free PMC article.
-
Bioremediation of mortar made from Ordinary Portland Cement degraded by Thiobacillus thioparus using Bacillus flexus.Heliyon. 2021 Jun 4;7(6):e07215. doi: 10.1016/j.heliyon.2021.e07215. eCollection 2021 Jun. Heliyon. 2021. PMID: 34159272 Free PMC article.
-
Impact of bacterial admixtures on the compressive and tensile strengths, permeability, and pore structure of ternary mortars: Comparative study of ureolytic and phototrophic bacteria.Biotechnol J. 2024 Jan;19(1):e2300157. doi: 10.1002/biot.202300157. Epub 2023 Oct 13. Biotechnol J. 2024. PMID: 37779350
-
Improved strength and durability of concrete through metabolic activity of ureolytic bacteria.Environ Sci Pollut Res Int. 2018 Aug;25(22):21451-21458. doi: 10.1007/s11356-017-9347-0. Epub 2017 Jun 7. Environ Sci Pollut Res Int. 2018. PMID: 28593545
-
Insights into the Current Trends in the Utilization of Bacteria for Microbially Induced Calcium Carbonate Precipitation.Materials (Basel). 2020 Nov 5;13(21):4993. doi: 10.3390/ma13214993. Materials (Basel). 2020. PMID: 33167607 Free PMC article. Review.
References
-
- Han P., Geng W.J., Li M.N., Jia S.R., Yin J.L., Xu X.R. Improvement of Biomineralization of Sporosarcina pasteurii as Biocementing Material for Concrete Repair by Atmospheric and Room Temperature Plasma Mutagenesis and Response Surface Methodology. J. Microbiol. Biotechnol. 2021;31:1311–1322. doi: 10.4014/jmb.2104.04019. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

