Identification of Exhaled Metabolites in Children with Cystic Fibrosis
- PMID: 36295881
- PMCID: PMC9611656
- DOI: 10.3390/metabo12100980
Identification of Exhaled Metabolites in Children with Cystic Fibrosis
Abstract
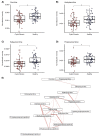
The early detection of inflammation and infection is important to prevent irreversible lung damage in cystic fibrosis. Novel and non-invasive monitoring tools would be of high benefit for the quality of life of patients. Our group previously detected over 100 exhaled mass-to-charge (m/z) features, using on-line secondary electrospray ionization high-resolution mass spectrometry (SESI-HRMS), which distinguish children with cystic fibrosis from healthy controls. The aim of this study was to annotate as many m/z features as possible with putative chemical structures. Compound identification was performed by applying a rigorous workflow, which included the analysis of on-line MS2 spectra and a literature comparison. A total of 49 discriminatory exhaled compounds were putatively identified. A group of compounds including glycolic acid, glyceric acid and xanthine were elevated in the cystic fibrosis group. A large group of acylcarnitines and aldehydes were found to be decreased in cystic fibrosis. The proposed compound identification workflow was used to identify signatures of volatile organic compounds that discriminate children with cystic fibrosis from healthy controls, which is the first step for future non-invasive and personalized applications.
Keywords: SESI-HRMS; breath analysis; children; cystic fibrosis; infection; inflammation; putative compound identification.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study, in the collection, analyses, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
Figures



References
-
- Laguna T.A., Wagner B.D., Williams C.B., Stevens M.J., Robertson C.E., Welchlin C.W., Moen C.E., Zemanick E.T., Harris J.K. Airway Microbiota in Bronchoalveolar Lavage Fluid from Clinically Well Infants with Cystic Fibrosis. PLoS ONE. 2016;11:e0167649. doi: 10.1371/journal.pone.0167649. - DOI - PMC - PubMed
-
- Singh K.D., Tancev G., Decrue F., Usemann J., Appenzeller R., Barreiro P., Jaumà G., Macia Santiago M., Vidal de Miguel G., Frey U., et al. Standardization Procedures for Real-Time Breath Analysis by Secondary Electrospray Ionization High-Resolution Mass Spectrometry. Anal. Bioanal. Chem. 2019;411:4883–4898. doi: 10.1007/s00216-019-01764-8. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

