Conquering Cancer Multi-Drug Resistance Using Curcumin and Cisplatin Prodrug-Encapsulated Mesoporous Silica Nanoparticles for Synergistic Chemo- and Photodynamic Therapies
- PMID: 36296885
- PMCID: PMC9609490
- DOI: 10.3390/nano12203693
Conquering Cancer Multi-Drug Resistance Using Curcumin and Cisplatin Prodrug-Encapsulated Mesoporous Silica Nanoparticles for Synergistic Chemo- and Photodynamic Therapies
Abstract
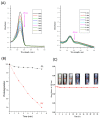
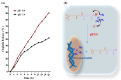
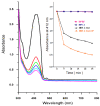
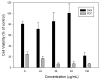
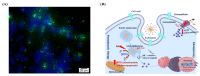
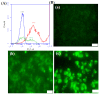
Recently, the development of anti-cancer approaches using different physical or chemical pathways has shifted from monotherapy to synergistic therapy, which can enhance therapeutic effects. As a result, enormous efforts have been devoted to developing various delivery systems encapsulated with dual agents for synergistic effects and to combat cancer cells acquired drug resistance. In this study, we show how to make Institute of Bioengineering and Nanotechnology (IBN)-1-based mesoporous silica nanoparticles (MSNs) for multifunctional drug delivery to overcome drug resistance cancer therapy. Initially, curcumin (Cur)-embedded IBN-1 nanocomposites (IBN-1-Cur) are synthesized in a simple one-pot co-condensation and then immobilized with the prodrug of Cisplatin (CP) on the carboxylate-modified surface (IBN-1-Cur-CP) to achieve photodynamic therapy (PDT) and chemotherapy in one platform, respectively, in the fight against multidrug resistance (MDR) of MES-SA/DX5 cancer cells. The Pluronic F127 triblock copolymer, as the structure-directing agent, in nanoparticles acts as a p-glycoprotein (p-gp) inhibitor. These designed hybrid nanocomposites with excellent structural properties are efficiently internalized by the endocytosis and successfully deliver Cur and CP molecules into the cytosol. Furthermore, the presence of Cur photosensitizer in the nanochannels of MSNs resulted in increased levels of cellular reactive oxygen species (ROS) under light irradiation. Thus, IBN-1-Cur-CP showed excellent anti-cancer therapy in the face of MES-SA/DX5 resistance cancer cells, owing to the synergistic effects of chemo- and photodynamic treatment.
Keywords: Cisplatin; mesoporous silica nanoparticles; p-glycoprotein; photodynamic therapy; reactive oxygen species.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Co-Delivery of Cisplatin Prodrug and Chlorin e6 by Mesoporous Silica Nanoparticles for Chemo-Photodynamic Combination Therapy to Combat Drug Resistance.ACS Appl Mater Interfaces. 2016 Jun 1;8(21):13332-40. doi: 10.1021/acsami.6b03881. Epub 2016 May 17. ACS Appl Mater Interfaces. 2016. PMID: 27164222
-
Co-Delivery of Cisplatin and Curcumin Using Mesoporous Silica Nanoparticles to Improve their Anticancer Effects.Pharm Nanotechnol. 2023;11(4):364-372. doi: 10.2174/2211738511666230327123627. Pharm Nanotechnol. 2023. PMID: 36974418
-
Enhanced photodynamic therapy of curcumin using biodegradable PLGA coated mesoporous silica nanoparticles.Eur J Pharm Biopharm. 2024 Nov;204:114503. doi: 10.1016/j.ejpb.2024.114503. Epub 2024 Sep 18. Eur J Pharm Biopharm. 2024. PMID: 39303950
-
Current trends in smart mesoporous silica-based nanovehicles for photoactivated cancer therapy.J Control Release. 2021 Nov 10;339:445-472. doi: 10.1016/j.jconrel.2021.10.005. Epub 2021 Oct 9. J Control Release. 2021. PMID: 34637819 Review.
-
Recent Advances in Photodynamic Therapy: Metal-Based Nanoparticles as Tools to Improve Cancer Therapy.Pharmaceutics. 2024 Jul 12;16(7):932. doi: 10.3390/pharmaceutics16070932. Pharmaceutics. 2024. PMID: 39065629 Free PMC article. Review.
Cited by
-
Recent Advances in Curcumin-Based Combination Nanomedicines for Cancer Therapy.J Funct Biomater. 2023 Aug 2;14(8):408. doi: 10.3390/jfb14080408. J Funct Biomater. 2023. PMID: 37623653 Free PMC article. Review.
-
Curcumin-loaded mesoporous silica nanoparticles for drug delivery: synthesis, biological assays and therapeutic potential - a review.RSC Adv. 2023 Jul 24;13(32):22250-22267. doi: 10.1039/d3ra02772d. eCollection 2023 Jul 19. RSC Adv. 2023. PMID: 37492509 Free PMC article. Review.
-
Recent Advances in Nanoparticle-Mediated Co-Delivery System: A Promising Strategy in Medical and Agricultural Field.Int J Mol Sci. 2023 Mar 7;24(6):5121. doi: 10.3390/ijms24065121. Int J Mol Sci. 2023. PMID: 36982200 Free PMC article. Review.
-
Resistance mechanisms of non-small cell lung cancer and improvement of treatment effects through nanotechnology: a narrative review.J Thorac Dis. 2024 Nov 30;16(11):8039-8052. doi: 10.21037/jtd-24-1078. Epub 2024 Nov 15. J Thorac Dis. 2024. PMID: 39678887 Free PMC article. Review.
-
Cisplatin-Based Combination Therapy for Enhanced Cancer Treatment.Curr Drug Targets. 2024;25(7):473-491. doi: 10.2174/0113894501294182240401060343. Curr Drug Targets. 2024. PMID: 38591210 Review.
References
-
- Danesi R., Innocenti F., Fogli S., Gennari A., Baldini E., Di Paolo A., Salvadori B., Bocci G., Conte P.F., Del Tacca M. Pharmacokinetics and pharmacodynamics of combination chemotherapy with paclitaxel and epirubicin in breast cancer patients. Br. J. Clin. Pharmacol. 2002;53:508–518. doi: 10.1046/j.1365-2125.2002.01579.x. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

