Molecular Imaging of Ultrasound-Mediated Blood-Brain Barrier Disruption in a Mouse Orthotopic Glioblastoma Model
- PMID: 36297663
- PMCID: PMC9610067
- DOI: 10.3390/pharmaceutics14102227
Molecular Imaging of Ultrasound-Mediated Blood-Brain Barrier Disruption in a Mouse Orthotopic Glioblastoma Model
Abstract
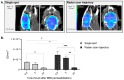
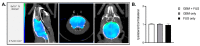
Glioblastoma (GBM) is an aggressive and malignant primary brain tumor. The blood-brain barrier (BBB) limits the therapeutic options available to tackle this incurable tumor. Transient disruption of the BBB by focused ultrasound (FUS) is a promising and safe approach to increase the brain and tumor concentration of drugs administered systemically. Non-invasive, sensitive, and reliable imaging approaches are required to better understand the impact of FUS on the BBB and brain microenvironment. In this study, nuclear imaging (SPECT/CT and PET/CT) was used to quantify neuroinflammation 48 h post-FUS and estimate the influence of FUS on BBB opening and tumor growth in vivo. BBB disruptions were performed on healthy and GBM-bearing mice (U-87 MG xenograft orthotopic model). The BBB recovery kinetics were followed and quantified by [99mTc]Tc-DTPA SPECT/CT imaging at 0.5 h, 3 h and 24 h post-FUS. The absence of neuroinflammation was confirmed by [18F]FDG PET/CT imaging 48 h post-FUS. The presence of the tumor and its growth were evaluated by [68Ga]Ga-RGD2 PET/CT imaging and post-mortem histological analysis, showing that tumor growth was not influenced by FUS. In conclusion, molecular imaging can be used to evaluate the time frame for systemic treatment combined with transient BBB opening and to test its efficacy over time.
Keywords: PET; SPECT; blood-brain barrier; drug delivery; focused ultrasound; glioblastoma.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures




References
LinkOut - more resources
Full Text Sources

