Third BNT162b2 Vaccine Booster Dose against SARS-CoV-2-Induced Antibody Response among Healthcare Workers
- PMID: 36298606
- PMCID: PMC9607254
- DOI: 10.3390/vaccines10101741
Third BNT162b2 Vaccine Booster Dose against SARS-CoV-2-Induced Antibody Response among Healthcare Workers
Abstract
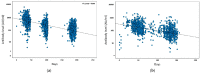
This study assessed humoral response to the third BNT162b2 dose among healthcare workers (HCW). This prospective cohort study of HCW tested for anti-spike antibodies (LIAISON SARS-CoV-2 S1/S2 IgG assay) at 1, 3, 6, 9, and 12 months after receiving the second BNT162b2 vaccine dose (tests 1, 2, 3, 4, and 5, respectively). A third (booster) vaccination dose was introduced before test 4. Linear regression model was used to determine the humoral response following vaccine doses. For each serology test, changes in log-transformed antibody concentrations over time, adjusted for age, sex, underlying diseases, steroid treatment, and smoking were described using the general linear mix model. Serology tests were performed at 3, 6, 9, and 12 months after the second vaccine dose in 1113, 1058, 986, and 939 participants, respectively. The third dose was received by 964 participants before the 9-month tests, 797 of whom participated in the 9- and 12-month serology tests. A significant inverse correlation was noted between time from third dose and antibody concentrations (Spearman correlation −0.395; p < 0.001). Age (p < 0.0001; CI 95% −0.005−−0.004), heart disease (p < 0.0001; CI 95% −0.177−−0.052), immunodeficiency (p < 0.0001; CI 95% 0.251−−0.106), and smoking (p < 0.0001; CI 95% −0.122−−0.040) were significantly associated with decreased antibody concentrations. Female sex (p = 0.03; CI 95% 0.013−0.066) was associated with increased antibody concentrations. The third booster dose had a better effect on immunogenicity, with higher antibody concentrations among tested HCW. Heart disease, smoking, and other known risk factors were associated with decreased antibody concentrations.
Keywords: BNT162b2 vaccine; SARS-CoV-2; antibody concentrations; booster; humoral response.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Our World in Data Website. Coronavirus Pandemic (COVID-19). 2020 (updated 14 June 2022) [(accessed on 15 September 2022)]. Available online: https://ourworldindata.org/coronavirus.
-
- Haas E.J., Angulo F.J., McLaughlin J.M., Anis E., Singer S.R., Khan F., Brooks N., Smaja M., Mircus G., Pan K., et al. Impact and effectiveness of mRNA BNT162b2 vaccine against SARS-CoV-2 infections and COVID-19 cases, hospitalisations, and deaths following a nationwide vaccination campaign in Israel: An observational study using national surveillance data. Lancet. 2021;397:1819–1829. doi: 10.1016/S0140-6736(21)00947-8. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous