The Performances of Three Commercially Available Assays for the Detection of SARS-CoV-2 Antibodies at Different Time Points Following SARS-CoV-2 Infection
- PMID: 36298751
- PMCID: PMC9610468
- DOI: 10.3390/v14102196
The Performances of Three Commercially Available Assays for the Detection of SARS-CoV-2 Antibodies at Different Time Points Following SARS-CoV-2 Infection
Abstract
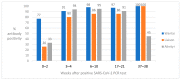
The aim of this study was to evaluate the performances of three commercially available antibody assays for the detection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) antibodies at different time points following SARS-CoV-2 infection. Sera from 536 cases, including 207 SARS-CoV-2 PCR positive, were tested for SARS-CoV-2 antibodies with the Wantai receptor binding domain (RBD) total antibody assay, Liaison S1/S2 IgG assay and Alinity i nucleocapsid IgG assay and compared to a two-step reference ELISA (SARS-CoV-2 RBD IgG and SARS-CoV-2 spike IgG). Diagnostic sensitivity, specificity, predictive values and Cohen's kappa were calculated for the commercial assays. The assay's sensitivities varied greatly, from 68.7% to 95.3%, but the specificities remained high (96.9-99.1%). The three tests showed good performances in sera sampled 31 to 60 days after PCR positivity compared to the reference ELISA. The total antibody test performed better than the IgG tests the first 30 days and the nucleocapsid IgG test showed reduced sensitivity two months or more after PCR positivity. Hence, the test performances at different time points should be taken into consideration in clinical practice and epidemiological studies. Spike or RBD IgG tests are preferable in sera sampled more than two months following SARS-CoV-2 infection.
Keywords: SARS-CoV-2; antibody assay; nucleocapsid; receptor binding domain; spike.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures
References
-
- Deeks J.J., Dinnes J., Takwoingi Y., Davenport C., Spijker R., Taylor-Phillips S., Adriano A., Beese S., Dretzke J., di Ruffano L.F., et al. Cochraine COVID-19 Diagnostic Test Accuracy Group. Antibody tests for identification of current and past infection with SARS-CoV-2. Cochrane Database Syst. Rev. 2020;6:Cd013652. - PMC - PubMed
-
- Grzelak L., Temmam S., Planchais C., Demeret C., Tondeur L., Huon C., Guivel-Benhassine F., Staropoli I., Chazal M., Dufloo J., et al. A comparison of four serological assays for detecting anti-SARS-CoV-2 antibodies in human serum samples from different populations. Sci. Transl. Med. 2020;2:eabc3103. doi: 10.1126/scitranslmed.abc3103. - DOI - PMC - PubMed
-
- Fjelltveit E.B., Blomberg B., Kuwelker K., Zhou F., Onyango T.B., Brokstad K.A., Elyanow R., Kaplan I.M., Tøndel C., Mohn K.G.I., et al. Bergen COVID-19 Research Group. Symptom burden and immune dynamics 6 to 18 months following mild SARS-CoV-2 infection—A case-control study. Clin. Infect. Dis. 2022:ciac655. doi: 10.1093/cid/ciac655. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

