Melatonin signalling in Schwann cells during neuroregeneration
- PMID: 36299487
- PMCID: PMC9589221
- DOI: 10.3389/fcell.2022.999322
Melatonin signalling in Schwann cells during neuroregeneration
Abstract
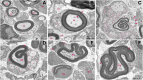
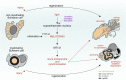
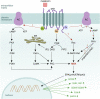
It has widely been thought that in the process of nerve regeneration Schwann cells populate the injury site with myelinating, non-myelinating, phagocytic, repair, and mesenchyme-like phenotypes. It is now clear that the Schwann cells modify their shape and basal lamina as to accommodate re-growing axons, at the same time clear myelin debris generated upon injury, and regulate expression of extracellular matrix proteins at and around the lesion site. Such a remarkable plasticity may follow an intrinsic functional rhythm or a systemic circadian clock matching the demands of accurate timing and precision of signalling cascades in the regenerating nervous system. Schwann cells react to changes in the external circadian clock clues and to the Zeitgeber hormone melatonin by altering their plasticity. This raises the question of whether melatonin regulates Schwann cell activity during neurorepair and if circadian control and rhythmicity of Schwann cell functions are vital aspects of neuroregeneration. Here, we have focused on different schools of thought and emerging concepts of melatonin-mediated signalling in Schwann cells underlying peripheral nerve regeneration and discuss circadian rhythmicity as a possible component of neurorepair.
Keywords: Schwann cells; basal lamina; circadian rhythm; extracellular matrix reorganisation; melatonin receptors; peripheral nerve injury.
Copyright © 2022 Klymenko and Lutz.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
-
- Altunkaynak B. Z., Delibaş B., Altun G., Deniz Ö. G. (2018). Melatonin and sciatic nerve injury repair: A current perspective. J. Neurorestoratology 6, 49–60. 10.2147/JN.S140614 - DOI
Publication types
LinkOut - more resources
Full Text Sources

