Effectiveness of combination therapy with ISA101 vaccine for the treatment of human papillomavirus-induced cervical cancer
- PMID: 36300095
- PMCID: PMC9589033
- DOI: 10.3389/fonc.2022.990877
Effectiveness of combination therapy with ISA101 vaccine for the treatment of human papillomavirus-induced cervical cancer
Abstract
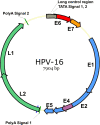
Cervical cancer is one of the women-associated tumors that affects numerous people yearly. It is the fourth most common malignancy in women worldwide. Following early diagnosis, this cancer can be cured mainly by traditional methods such as surgery, tumor resection, and chemotherapy; nonetheless, it becomes more challenging to treat in advanced and metastatic stages. With the advent of novel treatments such as angiogenesis inhibitors or immuno-checkpoint blockers in recent years, the survival rate of patients with advanced cervical cancer has significantly increased. However, it has not yet reached a satisfactory level. It has been revealed that human papillomavirus (HPV) infection is responsible for more than 90% of cervical cancer cases. However, evidence revealed that monotherapy with anti-HPV vaccines such as ISA101 could not affect tumor growth and progression in patients with HPV-induced cervical cancer. Therefore, combining ISA101 and immune checkpoint blockers or other immunotherapeutic approaches may be more robust and effective than monotherapy with ISA101 or immune checkpoint blockers for treating cervical cancer. This review summarizes the ISA101 properties, advantages and disadvantages. Furthermore, various conducted combination therapies with ISA101 and the effectiveness and challenges of this treatment have been discussed.
Keywords: ISA101; cervical cancer; human papilloma virus; immunotherapy; vaccine.
Copyright © 2022 Ding, Zhang, Zhang, Xu, Yu, Liang and Li.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Strong vaccine responses during chemotherapy are associated with prolonged cancer survival.Sci Transl Med. 2020 Mar 18;12(535):eaaz8235. doi: 10.1126/scitranslmed.aaz8235. Sci Transl Med. 2020. PMID: 32188726
-
Combining Immune Checkpoint Blockade and Tumor-Specific Vaccine for Patients With Incurable Human Papillomavirus 16-Related Cancer: A Phase 2 Clinical Trial.JAMA Oncol. 2019 Jan 1;5(1):67-73. doi: 10.1001/jamaoncol.2018.4051. JAMA Oncol. 2019. PMID: 30267032 Free PMC article. Clinical Trial.
-
Immunotherapeutic approaches for HPV-caused cervical cancer.Adv Protein Chem Struct Biol. 2022;129:51-90. doi: 10.1016/bs.apcsb.2021.11.002. Epub 2021 Dec 23. Adv Protein Chem Struct Biol. 2022. PMID: 35305725
-
Immunotherapy for human papillomavirus-associated disease and cervical cancer: review of clinical and translational research.J Gynecol Oncol. 2016 Sep;27(5):e51. doi: 10.3802/jgo.2016.27.e51. Epub 2016 May 31. J Gynecol Oncol. 2016. PMID: 27329199 Free PMC article. Review.
-
Cervical Cancer Immunotherapy: Facts and Hopes.Clin Cancer Res. 2021 Sep 15;27(18):4953-4973. doi: 10.1158/1078-0432.CCR-20-2833. Epub 2021 Apr 22. Clin Cancer Res. 2021. PMID: 33888488 Free PMC article. Review.
Cited by
-
Potential Therapeutic Targets for the Treatment of HPV-Associated Malignancies.Cancers (Basel). 2024 Oct 14;16(20):3474. doi: 10.3390/cancers16203474. Cancers (Basel). 2024. PMID: 39456568 Free PMC article. Review.
-
From Satirical Poems and Invisible Poisons to Radical Surgery and Organized Cervical Cancer Screening-A Historical Outline of Cervical Carcinoma and Its Relation to HPV Infection.Life (Basel). 2024 Feb 27;14(3):307. doi: 10.3390/life14030307. Life (Basel). 2024. PMID: 38541633 Free PMC article. Review.
-
Design and antiviral assessment of a panel of fusion proteins targeting human papillomavirus type 16.PLoS One. 2024 Oct 25;19(10):e0311137. doi: 10.1371/journal.pone.0311137. eCollection 2024. PLoS One. 2024. PMID: 39453911 Free PMC article.
-
Drugs and drug targets for the treatment of HPV-positive cervical cancer.Tumour Virus Res. 2025 Jun;19:200309. doi: 10.1016/j.tvr.2024.200309. Epub 2024 Dec 19. Tumour Virus Res. 2025. PMID: 39709045 Free PMC article. Review.
References
-
- Yagi A, Ueda Y, Kakuda M, Tanaka Y, Ikeda S, Matsuzaki S, et al. . Epidemiologic and clinical analysis of cervical cancer using data from the population-based Osaka cancer RegistryEpidemiologic and clinical analysis of cervical cancer. Cancer Res (2019) 79(6):1252–9. doi: 10.1158/0008-5472.CAN-18-3109 - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

