Enteral and Parenteral Treatment with Caffeine for Preterm Infants in the Delivery Room: A Randomised Trial
- PMID: 36301511
- PMCID: PMC9810558
- DOI: 10.1007/s40272-022-00541-y
Enteral and Parenteral Treatment with Caffeine for Preterm Infants in the Delivery Room: A Randomised Trial
Erratum in
-
Correction: Enteral and Parenteral Treatment with Caffeine for Preterm Infants in the Delivery Room: A Randomised Trial.Paediatr Drugs. 2024 Mar;26(2):215. doi: 10.1007/s40272-024-00619-9. Paediatr Drugs. 2024. PMID: 38363546 Free PMC article. No abstract available.
Abstract
Background: Early treatment with caffeine in the delivery room (DR) has been proposed to decrease the need for mechanical ventilation (MV) by limiting episodes of apnoea and improving respiratory mechanics in preterm infants. Our aim was to verify the hypothesis that intravenous or enteral administration of caffeine can be performed in the preterm infant in the DR.
Methods: Infants with 25±0-29±6 weeks of gestational age were enrolled and randomised to receive 20 mg/kg of caffeine citrate intravenously, via the umbilical vein, or enterally, through an orogastric tube, within 10 min of birth. Caffeine blood level was measured at 60 ± 15 min after administration and 60 ± 15 min before the next dose (5 mg/kg). The primary endpoint was evaluation of the success rate of intravenous and enteral administration of caffeine in the DR.
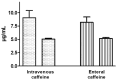
Results: Nineteen patients were treated with intravenous caffeine and 19 with enteral caffeine. In all patients the procedure was successfully performed. Peak blood level of caffeine 60 ± 15 min after administration in the DR was found to be below the therapeutic range (5 µg/mL) in 25 % of samples and above the therapeutic range in 3%. Blood level of caffeine 60 ± 15 min before administration of the second dose was found to be below the therapeutic range in 18% of samples.
Conclusions: Intravenous and enteral administration of caffeine can be performed in the DR without interfering with infants' postnatal assistance. Some patients did not reach the therapeutic range, raising the question of which dose is the most effective to prevent MV.
Clinical trial registration: ClinicalTrials.gov identifier NCT04044976; EudraCT number 2018-003626-91.
© 2022. The Author(s).
Conflict of interest statement
CD received honoraria from Chiesi Farmaceutici Spa and Vyaire Medical Inc. for scientific consultancy. Remaining authors declare that they have no competing interests.
Figures

References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

