Intestinal epithelial BLT1 promotes mucosal repair
- PMID: 36301666
- PMCID: PMC9746898
- DOI: 10.1172/jci.insight.162392
Intestinal epithelial BLT1 promotes mucosal repair
Abstract
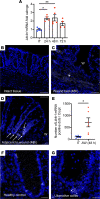
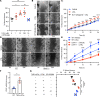
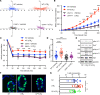
Acute and chronic intestinal inflammation is associated with epithelial damage, resulting in mucosal wounds in the forms of erosions and ulcers in the intestinal tract. Intestinal epithelial cells (IECs) and immune cells in the wound milieu secrete cytokines and lipid mediators to influence repair. Leukotriene B4 (LTB4), a lipid chemokine, binds to its receptor BLT1 and promotes migration of immune cells to sites of active inflammation; however, a role for intestinal epithelial BLT1 during mucosal wound repair is not known. Here we report that BLT1 was expressed in IECs both in vitro and in vivo, where it functioned as a receptor not only for LTB4 but also for another ligand, resolvin E1. Intestinal epithelial BLT1 expression was increased when epithelial cells were exposed to an inflammatory microenvironment. Using human and murine primary colonic epithelial cells, we reveal that the LTB4/BLT1 pathway promoted epithelial migration and proliferation leading to accelerated epithelial wound repair. Furthermore, in vivo intestinal wound repair experiments in BLT1-deficient mice and bone marrow chimeras demonstrated an important contribution of epithelial BLT1 during colonic mucosal wound repair. Taken together, our findings show a potentially novel prorepair in IEC mechanism mediated by BLT1 signaling.
Keywords: Gastroenterology; Inflammation; Inflammatory bowel disease.
Figures


Similar articles
-
Modulation of leukotriene B4 receptor 1 signaling by receptor for advanced glycation end products (RAGE).FASEB J. 2016 May;30(5):1811-22. doi: 10.1096/fj.201500117. Epub 2016 Jan 26. FASEB J. 2016. PMID: 26813973
-
Resolvin E1 is a pro-repair molecule that promotes intestinal epithelial wound healing.Proc Natl Acad Sci U S A. 2020 Apr 28;117(17):9477-9482. doi: 10.1073/pnas.1921335117. Epub 2020 Apr 16. Proc Natl Acad Sci U S A. 2020. PMID: 32300016 Free PMC article.
-
Macrophage LTB4 drives efficient phagocytosis of Borrelia burgdorferi via BLT1 or BLT2.J Lipid Res. 2017 Mar;58(3):494-503. doi: 10.1194/jlr.M068882. Epub 2017 Jan 4. J Lipid Res. 2017. PMID: 28053185 Free PMC article.
-
Identification, signaling, and functions of LTB4 receptors.Semin Immunol. 2017 Oct;33:30-36. doi: 10.1016/j.smim.2017.07.010. Semin Immunol. 2017. PMID: 29042026 Review.
-
Development, validation and implementation of an in vitro model for the study of metabolic and immune function in normal and inflamed human colonic epithelium.Dan Med J. 2015 Jan;62(1):B4973. Dan Med J. 2015. PMID: 25557335 Review.
Cited by
-
A mouse protozoan boosts antigen-specific mucosal IgA responses in a specific lipid metabolism- and signaling-dependent manner.Nat Commun. 2024 Sep 10;15(1):7914. doi: 10.1038/s41467-024-52336-z. Nat Commun. 2024. PMID: 39256385 Free PMC article.
-
Resolvins and cysteinyl-containing pro-resolving mediators activate resolution of infectious inflammation and tissue regeneration.Prostaglandins Other Lipid Mediat. 2023 Jun;166:106718. doi: 10.1016/j.prostaglandins.2023.106718. Epub 2023 Feb 21. Prostaglandins Other Lipid Mediat. 2023. PMID: 36813255 Free PMC article. Review.
-
Fatty acids and lipid mediators in inflammatory bowel disease: from mechanism to treatment.Front Immunol. 2023 Oct 5;14:1286667. doi: 10.3389/fimmu.2023.1286667. eCollection 2023. Front Immunol. 2023. PMID: 37868958 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

