A Bayesian model for chronic pain
- PMID: 36303889
- PMCID: PMC9595216
- DOI: 10.3389/fpain.2022.966034
A Bayesian model for chronic pain
Abstract
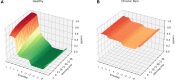
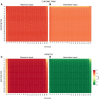
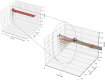
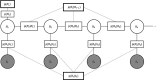
The perceiving mind constructs our coherent and embodied experience of the world from noisy, ambiguous and multi-modal sensory information. In this paper, we adopt the perspective that the experience of pain may similarly be the result of a probabilistic, inferential process. Prior beliefs about pain, learned from past experiences, are combined with incoming sensory information in a Bayesian manner to give rise to pain perception. Chronic pain emerges when prior beliefs and likelihoods are biased towards inferring pain from a wide range of sensory data that would otherwise be perceived as harmless. We present a computational model of interoceptive inference and pain experience. It is based on a Bayesian graphical network which comprises a hidden layer, representing the inferred pain state; and an observable layer, representing current sensory information. Within the hidden layer, pain states are inferred from a combination of priors , transition probabilities between hidden states and likelihoods of certain observations . Using variational inference and free-energy minimization, the model is able to learn from observations over time. By systematically manipulating parameter settings, we demonstrate that the model is capable of reproducing key features of both healthy- and chronic pain experience. Drawing on mathematical concepts, we finally simulate treatment resistant chronic pain and discuss mathematically informed treatment options.
Keywords: Bayesian inference; belief-propagation; chronic pain; computational psychiatry; free energy; graphical models; interoception; predictive coding.
© 2022 Eckert, Pabst and Endres DM.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




References
-
- IASP. Iasp terminology. (2017). Available at: https://www.iasp-pain.org/Education/Content.aspx?ItemNumber=1698navItemNumb
LinkOut - more resources
Full Text Sources
Miscellaneous

