Amyloid beta and its naturally occurring N-terminal variants are potent activators of human and mouse formyl peptide receptor 1
- PMID: 36309087
- PMCID: PMC9694488
- DOI: 10.1016/j.jbc.2022.102642
Amyloid beta and its naturally occurring N-terminal variants are potent activators of human and mouse formyl peptide receptor 1
Abstract
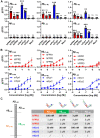
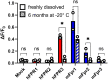
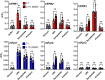
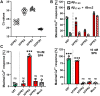
Formyl peptide receptors (FPRs) may contribute to inflammation in Alzheimer's disease through interactions with neuropathological Amyloid beta (Aβ) peptides. Previous studies reported activation of FPR2 by Aβ1-42, but further investigation of other FPRs and Aβ variants is needed. This study provides a comprehensive overview of the interactions of mouse and human FPRs with different physiologically relevant Aβ-peptides using transiently transfected cells in combination with calcium imaging. We observed that, in addition to hFPR2, all other hFPRs also responded to Aβ1-42, Aβ1-40, and the naturally occurring variants Aβ11-40 and Aβ17-40. Notably, Aβ11-40 and Aβ17-40 are very potent activators of mouse and human FPR1, acting at nanomolar concentrations. Buffer composition and aggregation state are extremely crucial factors that critically affect the interaction of Aβ with different FPR subtypes. To investigate the physiological relevance of these findings, we examined the effects of Aβ11-40 and Aβ17-40 on the human glial cell line U87. Both peptides induced a strong calcium flux at concentrations that are very similar to those obtained in experiments for hFPR1 in HEK cells. Further immunocytochemistry, qPCR, and pharmacological experiments verified that these responses were primarily mediated through hFPR1. Chemotaxis experiments revealed that Aβ11-40 but not Aβ17-40 evoked cell migration, which argues for a functional selectivity of different Aβ peptides. Together, these findings provide the first evidence that not only hFPR2 but also hFPR1 and hFPR3 may contribute to neuroinflammation in Alzheimer's disease through an interaction with different Aβ variants.
Keywords: Alzheimer's disease; amyloid beta; formyl peptide receptors; glia; neuroinflammation.
Copyright © 2022 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of interest The authors declare that they have no conflicts of interest with the contents of this article.
Figures









References
-
- AlzheimerAssociation 2019 Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2019;15:321–387.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

