Long-acting acid-sensitive ketal-linked dexamethasone microcrystals for treating experimental autoimmune uveitis
- PMID: 36313265
- PMCID: PMC9612960
- DOI: 10.1063/5.0118311
Long-acting acid-sensitive ketal-linked dexamethasone microcrystals for treating experimental autoimmune uveitis
Abstract
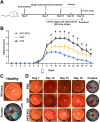
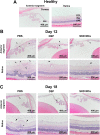
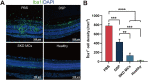
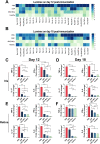
Corticosteroids have for some time been used as first-line drugs for the topical treatment of noninfectious uveitis, but poor ocular bioavailability and the rapid clearance of eye drops necessitate frequent dosing, reducing patient compliance. In this study, we used an acid-sensitive stearoxyl-ketal-dexamethasone pro-drug microcrystals (SKD MCs), which is consistently safe and effective in the control of uveitis inflammation in rats. We used a rat model of experimental autoimmune uveitis (EAU) to evaluate the effects of SKD MCs in terms of clinical manifestations, molecular biology, pathological histology, and visual electrophysiology compared to dexamethasone sodium phosphate injection or phosphate-buffered saline. SKD MCs significantly reduced inflammation in EAU, improved the ability to suppress inflammatory cytokines and to protect retinal function, and significantly reduced retinal microglia activation, with no increase in intraocular pressure throughout the treatment. Our results indicate that the SKD MCs formulation holds promise as a new strategy for the treatment of noninfectious uveitis and potentially other ocular inflammatory diseases.
© 2022 Author(s).
Figures


References
LinkOut - more resources
Full Text Sources

