Diagnostic biomarkers for active tuberculosis: progress and challenges
- PMID: 36314872
- PMCID: PMC9728055
- DOI: 10.15252/emmm.202114088
Diagnostic biomarkers for active tuberculosis: progress and challenges
Abstract
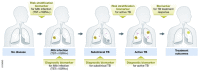
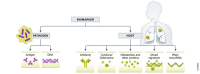
Tuberculosis (TB) is a leading cause of morbidity and mortality from a single infectious agent, despite being preventable and curable. Early and accurate diagnosis of active TB is critical to both enhance patient care, improve patient outcomes, and break Mycobacterium tuberculosis (Mtb) transmission cycles. In 2020 an estimated 9.9 million people fell ill from Mtb, but only a little over half (5.8 million) received an active TB diagnosis and treatment. The World Health Organization has proposed target product profiles for biomarker- or biosignature-based diagnostics using point-of-care tests from easily accessible specimens such as urine or blood. Here we review and summarize progress made in the development of pathogen- and host-based biomarkers for active TB diagnosis. We describe several unique patient populations that have posed challenges to development of a universal diagnostic TB biomarker, such as people living with HIV, extrapulmonary TB, and children. We also review additional limitations to widespread validation and utilization of published biomarkers. We conclude with proposed solutions to enhance TB diagnostic biomarker validation and uptake.
Keywords: active TB; biomarkers; diagnostic biomarkers; tuberculosis.
© 2022 The Authors. Published under the terms of the CC BY 4.0 license.
Figures


References
-
- Albuquerque VVS, Kumar NP, Fukutani KF, Vasconcelos B, Arriaga MB, Silveira‐Mattos PS, Babu S, Andrade BB (2019) Plasma levels of C‐reactive protein, matrix metalloproteinase‐7 and lipopolysaccharide‐binding protein distinguish active pulmonary or extrapulmonary tuberculosis from uninfected controls in children. Cytokine 123: 154773 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

