Polyphosphoric acid-promoted one-pot synthesis and neuroprotective effects of flavanones against NMDA-induced injury in PC12 cells
- PMID: 36320275
- PMCID: PMC9528730
- DOI: 10.1039/d2ra03562f
Polyphosphoric acid-promoted one-pot synthesis and neuroprotective effects of flavanones against NMDA-induced injury in PC12 cells
Abstract
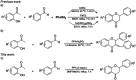
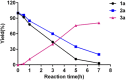
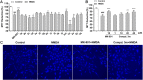
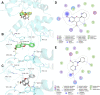
We report herein an efficient polyphosphoric acid (PPA) promoted one-pot protocol for the synthesis of flavanone derivatives from 2-hydroxyacetophenones and benzaldehydes. A variety of flavanones were produced in moderate to excellent yields and evaluated for their neuroprotective effects against N-methyl-d-aspartate (NMDA)-induced excitotoxicity in PC12 cells. Derivatives bearing electron-donating groups exhibited better neuroprotective activity. Compound 3m demonstrated the best protective potency and reversed the intracellular calcium (Ca2+) influx caused by NMDA, suggesting that flavanones protected the PC12 cells against NMDA-induced neurotoxicity via inhibition of Ca2+ overload.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures

Similar articles
-
Synthesis of novel 1-phenyl-benzopyrrolizidin-3-one derivatives and evaluation of their cytoneuroprotective effects against NMDA-induced injury in PC12 cells.Bioorg Med Chem. 2022 Apr 1;59:116675. doi: 10.1016/j.bmc.2022.116675. Epub 2022 Feb 18. Bioorg Med Chem. 2022. PMID: 35202968
-
Bryonolic Acid, a Triterpenoid, Protect Against N-methyl-d-Aspartate-Induced Neurotoxicity in PC12 Cells.Molecules. 2016 Mar 28;21(4):418. doi: 10.3390/molecules21040418. Molecules. 2016. PMID: 27043504 Free PMC article.
-
PFOS-induced excitotoxicity is dependent on Ca2+ influx via NMDA receptors in rat cerebellar granule neurons.Toxicol Appl Pharmacol. 2018 Oct 15;357:19-32. doi: 10.1016/j.taap.2018.08.015. Epub 2018 Aug 23. Toxicol Appl Pharmacol. 2018. PMID: 30144469
-
Pathologically-activated therapeutics for neuroprotection: mechanism of NMDA receptor block by memantine and S-nitrosylation.Curr Drug Targets. 2007 May;8(5):621-32. doi: 10.2174/138945007780618472. Curr Drug Targets. 2007. PMID: 17504105 Review.
-
Role of nonsynaptic GluN2B-containing NMDA receptors in excitotoxicity: evidence that fluoxetine selectively inhibits these receptors and may have neuroprotective effects.Brain Res Bull. 2013 Apr;93:32-8. doi: 10.1016/j.brainresbull.2012.10.005. Epub 2012 Oct 23. Brain Res Bull. 2013. PMID: 23089362 Review.
Cited by
-
Hydrogen Bond Assisted Three-Component Tandem Reactions to Access N-Alkyl-4-Quinolones.Molecules. 2023 Mar 2;28(5):2304. doi: 10.3390/molecules28052304. Molecules. 2023. PMID: 36903552 Free PMC article.
-
Polyphosphoric Acid-Promoted Efficient Synthesis of Cinnamides via Aldol Condensation of Amide.Molecules. 2024 Sep 29;29(19):4632. doi: 10.3390/molecules29194632. Molecules. 2024. PMID: 39407561 Free PMC article.
References
-
-
See for example:
- Siddikov G. U. Yuldashev M. P. Aripova S. F. Vdovin A. D. Abdullaev N. D. Botirov E. K. Chem. Nat. Compd. 2008;44:28.
- Zhou Z. Zhao P. Huang W. Yang G. Adv. Synth. Catal. 2006;348:63.
- Paredes A. Alzuru M. Mendez J. Rodríguez-Ortega M. Biol. Pharm. Bull. 2003;26:108. - PubMed
-
-
- Li W. D. Z. Ma B. C. Org. Lett. 2005;7:271. - PubMed
-
- Jung H. A. Jeong D. M. Chung H. Y. Lim H. A. Kim J. Y. Yoon N. Y. Choi J. S. Biol. Pharm. Bull. 2008;31:908. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

