Liver Histopathological Changes Related to Intraperitoneal Administration of Salicylic Acid/Fe3O4 Nanoparticles to C57BL/6 Mice
- PMID: 36320876
- PMCID: PMC9590356
- DOI: 10.12865/CHSJ.48.02.02
Liver Histopathological Changes Related to Intraperitoneal Administration of Salicylic Acid/Fe3O4 Nanoparticles to C57BL/6 Mice
Abstract
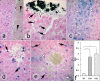
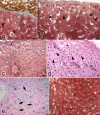
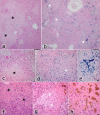
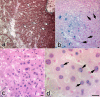
With a simple synthesis and easy engineering of physicochemical properties, iron oxide nanoparticles (IONPs) have become widely used in multiple biomedical applications. The study of IONPs toxicity has become an important issue, especially as the results reported so far are contradictory and range from lack of toxicity to cellular toxicity. The aim of this study was to evaluate the histopathological changes induced in mouse liver by long-term intraperitoneal injection of low doses of IONPs functionalized with salicylic acid (SaIONPs). The study was performed on C57BL/6 mice that received by intraperitoneal injection (IP), every two days, 0.6ml of SaIONPs aqueous suspension (35mg/kg body weight SaIONPs that contained 20mg/kg body weight of Fe3O4) for 28 days. The results of this study showed that the cumulative dose of 105mg/kg body weight SaIONPs (62mg/kg body weight of Fe3O4) induced histopathological changes in the subcapsular region of the mouse liver, possible by the release of salicylic acid into the peritoneal cavity. The cumulative dose of 244mg/kg body weight SaIONPs (145mg/kg body weight of Fe3O4) induced liver centrilobular necrosis, which requires the use of lower doses in biological applications. However, this may prove to be beneficial in the case of targeted accumulation of SaIONPs.
Keywords: Iron oxide nanoparticles; hidropic degeneration; rat liver; salicylic acid; toxicity.
Copyright © 2014, Medical University Publishing House Craiova.
Conflict of interest statement
None to declare.
Figures
References
LinkOut - more resources
Full Text Sources
