The effects of reduced nicotine content cigarettes on biomarkers of nicotine and toxicant exposure, smoking behavior and psychiatric symptoms in smokers with mood or anxiety disorders: A double-blind randomized trial
- PMID: 36322562
- PMCID: PMC9629593
- DOI: 10.1371/journal.pone.0275522
The effects of reduced nicotine content cigarettes on biomarkers of nicotine and toxicant exposure, smoking behavior and psychiatric symptoms in smokers with mood or anxiety disorders: A double-blind randomized trial
Abstract
Background: The U.S. Food and Drug Administration and the government of New Zealand have proposed a reduction of the nicotine content in cigarettes to very low levels. This study examined the potential effects of this regulation in smokers with affective disorders.
Methods: In a randomized controlled parallel group trial conducted at two sites in the USA (Penn State University, Hershey, PA and Massachusetts General Hospital, Boston, MA) 188 adult smokers with a current (n = 118) or lifetime (n = 70) anxiety or unipolar mood disorder, not planning to quit in the next 6 months, were randomly assigned (1:1) to smoke either Usual Nicotine Content (UNC) (11.6 mg nicotine/cigarette) research cigarettes, or Reduced Nicotine Content (RNC) research cigarettes where the nicotine content per cigarette was progressively reduced to 0.2 mg in five steps over 18 weeks. Participants were then offered the choice to either receive assistance to quit smoking, receive free research cigarettes, or resume using their own cigarette brand during a 12-week follow-up period. Main outcomes were biomarkers of nicotine and toxicant exposure, smoking behavior and dependence and severity of psychiatric symptoms. The pre-registered primary outcome was plasma cotinine.
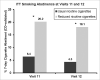
Results: A total of 143 (76.1%) randomized participants completed the randomized phase of the trial, 69 (73.4%) in the RNC group and 74 (78.8%) in the UNC group. After switching to the lowest nicotine content cigarettes, compared to smokers in the UNC group, at the last randomized visit the RNC group had significantly lower plasma cotinine (metabolite of nicotine): difference between groups, -175.7, 95% CI [-218.3, -133.1] ng/ml. Urine NNAL (metabolite of NNK, a lung carcinogen), exhaled carbon-monoxide, cigarette consumption, and cigarette dependence were also significantly lower in the RNC group than the UNC group. No between-group differences were found on a range of other biomarkers (e.g. 8-isoprostanes) or health indicators (e.g. blood pressure), or on 5 different psychiatric questionnaires, including the Kessler K6 measure of psychological distress. At the end of the subsequent 12-week treatment choice phase, those randomized to the RNC group were more likely to have quit smoking, based on initial intent-to-treat sample, n = 188 (18.1% RNC v 4.3% UNC, p = 0.004).
Conclusion: Reducing nicotine content in cigarettes to very low levels reduces some toxicant exposures and cigarette addiction and increases smoking cessation in smokers with mood and/or anxiety disorders, without worsening mental health.
Trial registration: TRN: NCT01928758, registered August 21, 2013.
Conflict of interest statement
JF has done paid consulting for pharmaceutical companies involved in producing smoking cessation medications, including GSK, Pfizer, Novartis, J&J, and Cypress Bioscience, and received a research grant from Pfizer Inc (not related to reduced nicotine cigarettes). AEE reports grant support to her institution from subcontracts from NIDA grants to Charles River Analytics and Brain Solutions LLC and consulting fees from Karuna Pharmaceuticals and Alkermes and editorial support from Pfizer for papers arising from the EAGLES trial. There are no competing interests to declare for other authors. This does not alter our adherence to PLOS ONE policies on sharing data and materials.
Figures



Similar articles
-
A two-site, two-arm, 34-week, double-blind, parallel-group, randomized controlled trial of reduced nicotine cigarettes in smokers with mood and/or anxiety disorders: trial design and protocol.BMC Public Health. 2017 Jan 19;17(1):100. doi: 10.1186/s12889-016-3946-4. BMC Public Health. 2017. PMID: 28103841 Free PMC article. Clinical Trial.
-
Reduced nicotine content cigarettes in smokers of low socioeconomic status: study protocol for a randomized control trial.Trials. 2017 Jul 3;18(1):300. doi: 10.1186/s13063-017-2038-9. Trials. 2017. PMID: 28673312 Free PMC article. Clinical Trial.
-
Switching to Progressively Reduced Nicotine Content Cigarettes in Smokers With Low Socioeconomic Status: A Double-Blind Randomized Clinical Trial.Nicotine Tob Res. 2021 May 24;23(6):992-1001. doi: 10.1093/ntr/ntaa247. Nicotine Tob Res. 2021. PMID: 33249498 Free PMC article. Clinical Trial.
-
Effect of menthol on nicotine reduction: Pooled results from two double-blind randomized controlled trials.Brain Res Bull. 2022 Oct 15;189:131-138. doi: 10.1016/j.brainresbull.2022.08.019. Epub 2022 Aug 28. Brain Res Bull. 2022. PMID: 36038015 Free PMC article. Review.
-
Behavioral and Subjective Effects of Reducing Nicotine in Cigarettes: A Cessation Commentary.Nicotine Tob Res. 2019 Dec 23;21(Suppl 1):S19-S21. doi: 10.1093/ntr/ntz100. Nicotine Tob Res. 2019. PMID: 31867644 Free PMC article. Review.
Cited by
-
Underreporting of non-study cigarette use by study participants confounds the interpretation of results from ambulatory clinical trial of reduced nicotine cigarettes.Harm Reduct J. 2024 Feb 8;21(1):35. doi: 10.1186/s12954-024-00953-8. Harm Reduct J. 2024. PMID: 38331789 Free PMC article.
-
Dietary Patterns among Smokers and Non-Smokers: Findings from the National Health and Nutritional Examination Survey (NHANES) 2017-2018.Nutrients. 2024 Jun 27;16(13):2035. doi: 10.3390/nu16132035. Nutrients. 2024. PMID: 38999783 Free PMC article.
-
What to Expect From a Low-Nicotine Product Standard for Cigarettes: A Response to "Denicotinized" Tobacco.JAMA. 2025 Feb 4;333(5):371-372. doi: 10.1001/jama.2024.23371. JAMA. 2025. PMID: 39652357
-
Perceptions and Interest in Lung Cancer Screening by Smoking Status: A Cross-Sectional Study of HINTS 6 (2022).Healthcare (Basel). 2024 Sep 30;12(19):1952. doi: 10.3390/healthcare12191952. Healthcare (Basel). 2024. PMID: 39408132 Free PMC article.
-
Cytisinicline to Speed Smoking Cessation in the United States.JAMA. 2023 Jul 11;330(2):129-130. doi: 10.1001/jama.2023.5939. JAMA. 2023. PMID: 37432440 Free PMC article. No abstract available.
References
-
- Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health. The Health Consequences of Smoking-50 Years of Progress: A Report of the Surgeon General. Reports of the Surgeon General. Atlanta (GA) 2014.
-
- Food and Drug Administration. FDA Announces Plans for Proposed Rule to Reduce Addictiveness of Cigarettes and Other Combusted Tobacco Products 2022. June 21 [cited 2022 August 29]. Available from: https://www.fda.gov/news-events/press-announcements/fda-announces-plans-....
-
- New Zealand Ministry of Health. Smokefree Aotearoa 2025 Action Plan—Auahi Kore Aotearoa Mahere Rautaki 2025. In: Ministry of Health, editor. Wellington: New Zealand Ministry of Health; 2021.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

