Pathogenicity and virulence of Mycobacterium leprae
- PMID: 36326715
- PMCID: PMC9635560
- DOI: 10.1080/21505594.2022.2141987
Pathogenicity and virulence of Mycobacterium leprae
Abstract
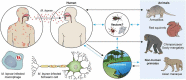
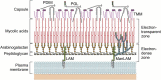
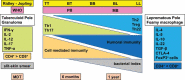
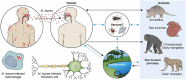
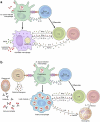
Leprosy is caused by Mycobacterium leprae (M. leprae) and M. lepromatosis, an obligate intracellular organism, and over 200,000 new cases occur every year. M. leprae parasitizes histiocytes (skin macrophages) and Schwann cells in the peripheral nerves. Although leprosy can be treated by multidrug therapy, some patients relapse or have a prolonged clinical course and/or experience leprosy reaction. These varying outcomes depend on host factors such as immune responses against bacterial components that determine a range of symptoms. To understand these host responses, knowledge of the mechanisms by which M. leprae parasitizes host cells is important. This article describes the characteristics of leprosy through bacteriology, genetics, epidemiology, immunology, animal models, routes of infection, and clinical findings. It also discusses recent diagnostic methods, treatment, and measures according to the World Health Organization (WHO), including prevention. Recently, the antibacterial activities of anti-hyperlipidaemia agents against other pathogens, such as M. tuberculosis and Staphylococcus aureus have been investigated. Our laboratory has been focused on the metabolism of lipids which constitute the cell wall of M. leprae. Our findings may be useful for the development of future treatments.
Keywords: Leprosy; Mycobacterium leprae; Schwann cell; lipids metabolism; macrophage; pseudogene.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures

References
-
- World Health Organization . Global leprosy (Hansen disease) update, 2019: time to step-up prevention initiatives. Wkly Epidemiol Rec. 2020;95(36):417–440.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
