Base-Mediated Nitrophenyl Reductive Cyclization for the Synthesis of Hexahydro-2,6-methano-1-benzazocines
- PMID: 36329560
- PMCID: PMC9680033
- DOI: 10.1021/acs.joc.2c02205
Base-Mediated Nitrophenyl Reductive Cyclization for the Synthesis of Hexahydro-2,6-methano-1-benzazocines
Abstract
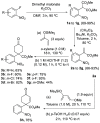
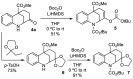
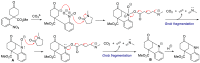
A Diels-Alder reaction leading to 4-nitrophenylcyclohexanones followed by a newly developed base-mediated reductive cyclization of the resulting ketone tethered to the nitrobenzene moiety gives access to the hexahydro-2,6-methano-1-benzazocine ring system present in several biologically interesting natural products such as aspernomine. The scope of the reaction was explored with eight substituted nitrobenzenes, obtaining yields of up to 87%. The highest cytotoxicity was observed in benzazocine 4h, bearing an enone moiety, which was active against eight cancer cell lines.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



References
-
- Gloer J. B. Antiinsectan natural products from fungal sclerotia. Acc. Chem. Res. 1995, 28, 343–350. 10.1021/ar00056a004. - DOI
-
- Gloer J. B.; Rinderknecht B. L.; Wicklow D. T.; Dowd P. F. Nominine: a new insecticidal indole diterpene from the sclerotia of Aspergillus nomius. J. Org. Chem. 1989, 54, 2530–2532. 10.1021/jo00272a012. - DOI
-
- Bian M.; Wang Z.; Xiong X.; Sun Y.; Matera C.; Nicolaou K. C.; Li A. Total Syntheses of Anominine and Tubingensin A. J. Am. Chem. Soc. 2012, 134, 8078–8081. 10.1021/ja302765m. - DOI - PubMed
- Goetz A. E.; Silberstein A. L.; Corsello M. A.; Garg N. K. Concise Enantiospecific Total Synthesis of Tubingensin A. J. Am. Chem. Soc. 2014, 136, 3036–3039. 10.1021/ja501142e. - DOI - PMC - PubMed
- Lu Z.; Li H.; Bian M.; Li A. Total Synthesis of Epoxyeujindole A. J. Am. Chem. Soc. 2015, 137, 13764–13767. 10.1021/jacs.5b09198. - DOI - PubMed
- Li H.; Chen Q.; Lu Z.; Li A. Total Syntheses of Aflavazole and 1,4-Hydroxyaflavinine. J. Am. Chem. Soc. 2016, 138, 15555–15558. 10.1021/jacs.6b10880. - DOI - PubMed
- Corsello M. A.; Kim J.; Garg N. K. Total synthesis of (−)-tubingensin B enabled by the strategic use of an aryne cyclization. Nature Chemistry. 2017, 9, 944–949. 10.1038/nchem.2801. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

