Empagliflozin in Patients with Chronic Kidney Disease
- PMID: 36331190
- PMCID: PMC7614055
- DOI: 10.1056/NEJMoa2204233
Empagliflozin in Patients with Chronic Kidney Disease
Abstract
Background: The effects of empagliflozin in patients with chronic kidney disease who are at risk for disease progression are not well understood. The EMPA-KIDNEY trial was designed to assess the effects of treatment with empagliflozin in a broad range of such patients.
Methods: We enrolled patients with chronic kidney disease who had an estimated glomerular filtration rate (eGFR) of at least 20 but less than 45 ml per minute per 1.73 m2 of body-surface area, or who had an eGFR of at least 45 but less than 90 ml per minute per 1.73 m2 with a urinary albumin-to-creatinine ratio (with albumin measured in milligrams and creatinine measured in grams) of at least 200. Patients were randomly assigned to receive empagliflozin (10 mg once daily) or matching placebo. The primary outcome was a composite of progression of kidney disease (defined as end-stage kidney disease, a sustained decrease in eGFR to <10 ml per minute per 1.73 m2, a sustained decrease in eGFR of ≥40% from baseline, or death from renal causes) or death from cardiovascular causes.
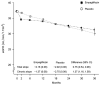
Results: A total of 6609 patients underwent randomization. During a median of 2.0 years of follow-up, progression of kidney disease or death from cardiovascular causes occurred in 432 of 3304 patients (13.1%) in the empagliflozin group and in 558 of 3305 patients (16.9%) in the placebo group (hazard ratio, 0.72; 95% confidence interval [CI], 0.64 to 0.82; P<0.001). Results were consistent among patients with or without diabetes and across subgroups defined according to eGFR ranges. The rate of hospitalization from any cause was lower in the empagliflozin group than in the placebo group (hazard ratio, 0.86; 95% CI, 0.78 to 0.95; P = 0.003), but there were no significant between-group differences with respect to the composite outcome of hospitalization for heart failure or death from cardiovascular causes (which occurred in 4.0% in the empagliflozin group and 4.6% in the placebo group) or death from any cause (in 4.5% and 5.1%, respectively). The rates of serious adverse events were similar in the two groups.
Conclusions: Among a wide range of patients with chronic kidney disease who were at risk for disease progression, empagliflozin therapy led to a lower risk of progression of kidney disease or death from cardiovascular causes than placebo. (Funded by Boehringer Ingelheim and others; EMPA-KIDNEY ClinicalTrials.gov number, NCT03594110; EudraCT number, 2017-002971-24.).
Copyright © 2022 Massachusetts Medical Society.
Figures


Comment in
-
Kidney benefits of SLGT2 inhibitors: evidence from clinical trials.Nat Rev Nephrol. 2023 Jan;19(1):3. doi: 10.1038/s41581-022-00659-9. Nat Rev Nephrol. 2023. PMID: 36450917 No abstract available.
-
Chronic Kidney Disease - Another Step Forward.N Engl J Med. 2023 Jan 12;388(2):179-180. doi: 10.1056/NEJMe2215286. N Engl J Med. 2023. PMID: 36630627 No abstract available.
-
In CKD, once-daily empagliflozin reduced progression of kidney disease or CV death at 2 y.Ann Intern Med. 2023 Mar;176(3):JC26. doi: 10.7326/J23-0009. Epub 2023 Mar 7. Ann Intern Med. 2023. PMID: 36877974
-
Niereninsuffizienz: Breiter Schutz durch SGLT2-Hemmer.MMW Fortschr Med. 2023 Apr;165(7):26-27. doi: 10.1007/s15006-023-2531-x. MMW Fortschr Med. 2023. PMID: 37016221 German. No abstract available.
-
Empagliflozin in Patients with Chronic Kidney Disease.N Engl J Med. 2023 Jun 15;388(24):2300. doi: 10.1056/NEJMc2301923. N Engl J Med. 2023. PMID: 37314720 No abstract available.
-
Empagliflozin in Patients with Chronic Kidney Disease.N Engl J Med. 2023 Jun 15;388(24):2300-2301. doi: 10.1056/NEJMc2301923. N Engl J Med. 2023. PMID: 37314721 No abstract available.
-
Empagliflozin in Patients with Chronic Kidney Disease.N Engl J Med. 2023 Jun 15;388(24):2301. doi: 10.1056/NEJMc2301923. N Engl J Med. 2023. PMID: 37314722 No abstract available.
-
Empagliflozin in Patients with Chronic Kidney Disease. Reply.N Engl J Med. 2023 Jun 15;388(24):2301-2302. doi: 10.1056/NEJMc2301923. N Engl J Med. 2023. PMID: 37314723 No abstract available.
References
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
