Moisture Sorption Isotherms of Sweet Cherry (Prunus Avium L.): Comparative Study of Kinetics and Thermodynamic Modeling of Five Varieties
- PMID: 36337164
- PMCID: PMC9629944
- DOI: 10.1155/2022/6786590
Moisture Sorption Isotherms of Sweet Cherry (Prunus Avium L.): Comparative Study of Kinetics and Thermodynamic Modeling of Five Varieties
Abstract
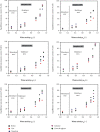
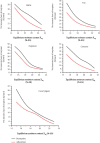
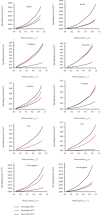
Moisture sorption isotherms of five sweet cherry cultivars (Prunus avium L.) at three temperatures of 30°C, 40°C, and 50°C, and water activity range of 0.057-0.898 were determined using the static gravimetric method. The sorption isotherms of all cultivars decreased with increasing temperature, and they all exhibited type II behavior according to the classification of IUPAC (International Union of Pure and Applied Chemistry). The isosteric heat of sorption, differential entropy, spreading pressure, and water surface area were determined, and the energy associated with the sorption processes was defined. The curves were fitted to GAB, PELEG, and ENDERBY models, and the GAB model gave the best fit for the whole set of data. The enthalpy-entropy compensation proved that the process occurs spontaneously and is fully controlled the enthalpy. The spreading pressure value varied with temperature in all sweet cherry cultivars in both the desorption and adsorption processes. The average surface area varied from 78.05 to 214.02 m2/g for desorption and from 49.0 to 204.4 m2/g for adsorption from 30 to 50°C.
Copyright © 2022 Rachida Ouaabou et al.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Ouaabou R., Ennahli S., Nabil B., et al. Multivariate cherry quality assessment using morphological, biochemical and volatile compound traits. International Journal of Fruit Science . 2020;20(sup3):S1328–S1347. doi: 10.1080/15538362.2020.1785988. - DOI
-
- Öztürk B., Ağlar E., Karakaya O., Saracoğlu O., Gün S. Effects of preharvest GA3, CaCl2 and modified atmosphere packaging treatments on specific phenolic compounds of sweet cherry. Turkish Journal of Food and Agriculture Sciences . 2020;2020:2687–3818. doi: 10.14744/turkjfas.2019.009. - DOI
-
- Mozetič B., Simčič M., Trebše P. Anthocyanins and hydroxycinnamic acids of Lambert Compact cherries (Prunus avium L.) after cold storage and 1-methylcyclopropene treatment. Food Chemistry . 2006;97(2):302–309. doi: 10.1016/j.foodchem.2005.04.018. - DOI
LinkOut - more resources
Full Text Sources

