Ciliated epithelia are key elements in the recruitment of bacterial partners in the squid-vibrio symbiosis
- PMID: 36340026
- PMCID: PMC9632347
- DOI: 10.3389/fcell.2022.974213
Ciliated epithelia are key elements in the recruitment of bacterial partners in the squid-vibrio symbiosis
Abstract
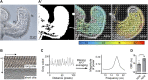
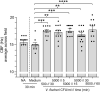
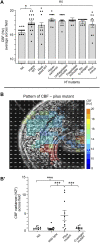
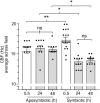
The Hawaiian bobtail squid, Euprymna scolopes, harvests its luminous symbiont, Vibrio fischeri, from the surrounding seawater within hours of hatching. During embryogenesis, the host animal develops a nascent light organ with ciliated fields on each lateral surface. We hypothesized that these fields function to increase the efficiency of symbiont colonization of host tissues. Within minutes of hatching from the egg, the host's ciliated fields shed copious amounts of mucus in a non-specific response to bacterial surface molecules, specifically peptidoglycan (PGN), from the bacterioplankton in the surrounding seawater. Experimental manipulation of the system provided evidence that nitric oxide in the mucus drives an increase in ciliary beat frequency (CBF), and exposure to even small numbers of V. fischeri cells for short periods resulted in an additional increase in CBF. These results indicate that the light-organ ciliated fields respond specifically, sensitively, and rapidly, to the presence of nonspecific PGN as well as symbiont cells in the ambient seawater. Notably, the study provides the first evidence that this induction of an increase in CBF occurs as part of a thus far undiscovered initial phase in colonization of the squid host by its symbiont, i.e., host recognition of V. fischeri cues in the environment within minutes. Using a biophysics-based mathematical analysis, we showed that this rapid induction of increased CBF, while accelerating bacterial advection, is unlikely to be signaled by V. fischeri cells interacting directly with the organ surface. These overall changes in CBF were shown to significantly impact the efficiency of V. fischeri colonization of the host organ. Further, once V. fischeri has fully colonized the host tissues, i.e., about 12-24 h after initial host-symbiont interactions, the symbionts drove an attenuation of mucus shedding from the ciliated fields, concomitant with an attenuation of the CBF. Taken together, these findings offer a window into the very first interactions of ciliated surfaces with their coevolved microbial partners.
Keywords: Euprymna scolopes; Vibrio fischeri; ciliary beat frequency; ciliated fields; light organ; symbiont harvesting.
Copyright © 2022 Gundlach, Nawroth, Kanso, Nasrin, Ruby and McFall-Ngai.
Conflict of interest statement
JN was employed by Helmholtz Zentrum München. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Persistent Interactions with Bacterial Symbionts Direct Mature-Host Cell Morphology and Gene Expression in the Squid-Vibrio Symbiosis.mSystems. 2018 Oct 2;3(5):e00165-18. doi: 10.1128/mSystems.00165-18. eCollection 2018 Sep-Oct. mSystems. 2018. PMID: 30320217 Free PMC article.
-
A peptidoglycan-recognition protein orchestrates the first steps of symbiont recruitment in the squid-vibrio symbiosis.Symbiosis. 2022 May;87(1):31-43. doi: 10.1007/s13199-022-00855-y. Epub 2022 Aug 6. Symbiosis. 2022. PMID: 36177150 Free PMC article.
-
Roles of Vibrio fischeri and nonsymbiotic bacteria in the dynamics of mucus secretion during symbiont colonization of the Euprymna scolopes light organ.Appl Environ Microbiol. 2002 Oct;68(10):5113-22. doi: 10.1128/AEM.68.10.5113-5122.2002. Appl Environ Microbiol. 2002. PMID: 12324362 Free PMC article.
-
The Role of Hemocytes in the Hawaiian Bobtail Squid, Euprymna scolopes: A Model Organism for Studying Beneficial Host-Microbe Interactions.Front Microbiol. 2017 Jan 6;7:2013. doi: 10.3389/fmicb.2016.02013. eCollection 2016. Front Microbiol. 2017. PMID: 28111565 Free PMC article. Review.
-
Nocturnal Acidification: A Coordinating Cue in the Euprymna scolopes-Vibrio fischeri Symbiosis.Int J Mol Sci. 2022 Mar 29;23(7):3743. doi: 10.3390/ijms23073743. Int J Mol Sci. 2022. PMID: 35409100 Free PMC article. Review.
Cited by
-
LitR and its quorum-sensing regulators modulate biofilm formation by Vibrio fischeri.J Bacteriol. 2025 Feb 20;207(2):e0047624. doi: 10.1128/jb.00476-24. Epub 2025 Jan 29. J Bacteriol. 2025. PMID: 39878466 Free PMC article.
-
An acidic microenvironment produced by the V-type ATPase of Euprymna scolopes promotes specificity during Vibrio fischeri recruitment.Commun Biol. 2024 Dec 18;7(1):1642. doi: 10.1038/s42003-024-07348-2. Commun Biol. 2024. PMID: 39695176 Free PMC article.
-
A bacterial membrane-disrupting protein stimulates animal metamorphosis.mBio. 2025 Feb 5;16(2):e0357324. doi: 10.1128/mbio.03573-24. Epub 2024 Dec 27. mBio. 2025. PMID: 39727418 Free PMC article.
References
-
- Brown B. E., Bythell J. C. (2005). Perspectives on mucus secretion in reef corals. Mar. Ecol. Prog. Ser. 296, 291–309. 10.3354/meps296291 - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

