PDGF-BB signaling via PDGFR-β regulates the maturation of blood vessels generated upon vasculogenic differentiation of dental pulp stem cells
- PMID: 36340037
- PMCID: PMC9627550
- DOI: 10.3389/fcell.2022.977725
PDGF-BB signaling via PDGFR-β regulates the maturation of blood vessels generated upon vasculogenic differentiation of dental pulp stem cells
Abstract
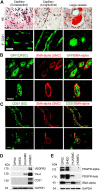
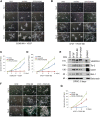
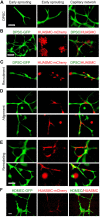
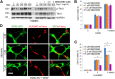
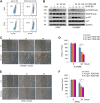
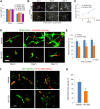
A functional vascular network requires that blood vessels are invested by mural cells. We have shown that dental pulp stem cells (DPSC) can undergo vasculogenic differentiation, and that the resulting vessels anastomize with the host vasculature and become functional (blood carrying) vessels. However, the mechanisms underlying the maturation of DPSC-derived blood vessels remains unclear. Here, we performed a series of studies to understand the process of mural cell investment of blood vessels generated upon vasculogenic differentiation of dental pulp stem cells. Primary human DPSC were co-cultured with primary human umbilical artery smooth muscle cells (HUASMC) in 3D gels in presence of vasculogenic differentiation medium. We observed DPSC capillary sprout formation and SMC recruitment, alignment and remodeling that resulted in complex vascular networks. While HUASMC enhanced the number of capillary sprouts and stabilized the capillary network when co-cultured with DPSC, HUASMC by themselves were unable to form capillary sprouts. In vivo, GFP transduced human DPSC seeded in biodegradable scaffolds and transplanted into immunodeficient mice generated functional human blood vessels invested with murine smooth muscle actin (SMA)-positive, GFP-negative cells. Inhibition of PDGFR-β signaling prevented the SMC investment of DPSC-derived capillary sprouts in vitro and of DPSC-derived blood vessels in vivo. In contrast, inhibition of Tie-2 signaling did not have a significant effect on the SMC recruitment in DPSC-derived vascular structures. Collectively, these results demonstrate that PDGF-BB signaling via PDGFR-β regulates the process of maturation (mural investment) of blood vessels generated upon vasculogenic differentiation of human dental pulp stem cells.
Keywords: angiogenesis; pericytes; self-renewal; smooth muscle cells; tissue regeneration; vasculogenesis.
Copyright © 2022 Zhang, Warner, Mantesso and Nör.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

