Tumor microenvironment-mediated immune tolerance in development and treatment of gastric cancer
- PMID: 36341377
- PMCID: PMC9630479
- DOI: 10.3389/fimmu.2022.1016817
Tumor microenvironment-mediated immune tolerance in development and treatment of gastric cancer
Abstract
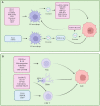
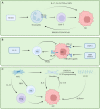
Tumor microenvironment is the general term for all non-cancer components and their metabolites in tumor tissue. These components include the extracellular matrix, fibroblasts, immune cells, and endothelial cells. In the early stages of tumors, the tumor microenvironment has a tumor suppressor function. As the tumor progresses, tumor immune tolerance is induced under the action of various factors, such that the tumor suppressor microenvironment is continuously transformed into a tumor-promoting microenvironment, which promotes tumor immune escape. Eventually, tumor cells manifest the characteristics of malignant proliferation, invasion, metastasis, and drug resistance. In recent years, stress effects of the extracellular matrix, metabolic and phenotypic changes of innate immune cells (such as neutrophils, mast cells), and adaptive immune cells in the tumor microenvironment have been revealed to mediate the emerging mechanisms of immune tolerance, providing us with a large number of emerging therapeutic targets to relieve tumor immune tolerance. Gastric cancer is one of the most common digestive tract malignancies worldwide, whose mortality rate remains high. According to latest guidelines, the first-line chemotherapy of advanced gastric cancer is the traditional platinum and fluorouracil therapy, while immunotherapy for gastric cancer is extremely limited, including only Human epidermal growth factor receptor 2 (HER-2) and programmed death ligand 1 (PD-L1) targeted drugs, whose benefits are limited. Clinical experiments confirmed that cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), vascular endothelial growth factor receptor (VEGFR) and other targeted drugs alone or in combination with other drugs have limited efficacy in patients with advanced gastric cancer, far less than in lung cancer, colon cancer, and other tumors. The failure of immunotherapy is mainly related to the induction of immune tolerance in the tumor microenvironment of gastric cancer. Therefore, solving the immune tolerance of tumors is key to the success of gastric cancer immunotherapy. In this study, we summarize the latest mechanisms of various components of the tumor microenvironment in gastric cancer for inducing immune tolerance and promoting the formation of the malignant phenotype of gastric cancer, as well as the research progress of targeting the tumor microenvironment to overcome immune tolerance in the treatment of gastric cancer.
Keywords: gastric cancer; immune tolerance; immunotherapy; tumor microenvironment; tumor-infiltrating immune cells.
Copyright © 2022 Liu, Li, Lu, Liu and Yang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Janjigian YY, Shitara K, Moehler M, Garrido M, Salman P, Shen L, et al. . First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (Checkmate 649): A randomised, open-label, phase 3 trial. Lancet (2021) 398(10294):27–40. doi: 10.1016/S0140-6736(21)00797-2 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

