Nutrient Availability and Biofilm Polysaccharide Shape the Bacillaene-Dependent Antagonism of Bacillus subtilis against Salmonella Typhimurium
- PMID: 36342318
- PMCID: PMC9769773
- DOI: 10.1128/spectrum.01836-22
Nutrient Availability and Biofilm Polysaccharide Shape the Bacillaene-Dependent Antagonism of Bacillus subtilis against Salmonella Typhimurium
Abstract
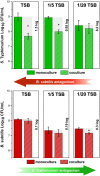
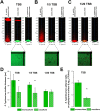
Salmonella enterica is one of the most common foodborne pathogens and, due to the spread of antibiotic resistance, new antimicrobial strategies are urgently needed to control it. In this study, we explored the probiotic potential of Bacillus subtilis PS-216 and elucidated the mechanisms that underlie the interactions between this soil isolate and the model pathogenic strain S. Typhimurium SL1344. The results reveal that B. subtilis PS-216 inhibits the growth and biofilm formation of S. Typhimurium through the production of the pks cluster-dependent polyketide bacillaene. The presence of S. Typhimurium enhanced the activity of the PpksC promoter that controls bacillaene production, suggesting that B. subtilis senses and responds to Salmonella. The level of Salmonella inhibition, overall PpksC activity, and PpksC induction by Salmonella were all higher in nutrient-rich conditions than in nutrient-depleted conditions. Although eliminating the extracellular polysaccharide production of B. subtilis via deletion of the epsA-O operon had no significant effect on inhibitory activity against Salmonella in nutrient-rich conditions, this deletion mutant showed an enhanced antagonism against Salmonella in nutrient-depleted conditions, revealing an intricate relationship between exopolysaccharide production, nutrient availability, and bacillaene synthesis. Overall, this work provides evidence on the regulatory role of nutrient availability, sensing of the competitor, and EpsA-O polysaccharide in the social outcome of bacillaene-dependent competition between B. subtilis and S. Typhimurium. IMPORTANCE Probiotic bacteria represent an alternative for controlling foodborne disease caused by Salmonella enterica, which constitutes a serious concern during food production due to its antibiotic resistance and resilience to environmental stress. Bacillus subtilis is gaining popularity as a probiotic, but its behavior in biofilms with pathogens such as Salmonella remains to be elucidated. Here, we show that the antagonism of B. subtilis is mediated by the polyketide bacillaene and that the production of bacillaene is a highly dynamic trait which depends on environmental factors such as nutrient availability and the presence of competitors. Moreover, the production of extracellular polysaccharides by B. subtilis further alters the influence of these factors. Hence, this work highlights the inhibitory effect of B. subtilis, which is condition-dependent, and the importance of evaluating probiotic strains under conditions relevant to the intended use.
Keywords: Bacillus subtilis; Salmonella enterica; biofilm; enteric pathogen; microbial competition; nutrients; polysaccharides; probiotics.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Bacillus subtilis ensures high spore quality in competition with Salmonella Typhimurium via the SigB-dependent pathway.ISME J. 2025 Jan 2;19(1):wraf052. doi: 10.1093/ismejo/wraf052. ISME J. 2025. PMID: 40098255 Free PMC article.
-
Bacillus subtilis Intraspecies Interactions Shape Probiotic Activity Against Salmonella Typhimurium.Microb Biotechnol. 2024 Dec;17(12):e70065. doi: 10.1111/1751-7915.70065. Microb Biotechnol. 2024. PMID: 39718437 Free PMC article.
-
Impact of Bacillus subtilis Antibiotic Bacilysin and Campylobacter jejuni Efflux Pumps on Pathogen Survival in Mixed Biofilms.Microbiol Spectr. 2022 Aug 31;10(4):e0215622. doi: 10.1128/spectrum.02156-22. Epub 2022 Aug 8. Microbiol Spectr. 2022. PMID: 35938811 Free PMC article.
-
Bacillaene Mediates the Inhibitory Effect of Bacillus subtilis on Campylobacter jejuni Biofilms.Appl Environ Microbiol. 2021 May 26;87(12):e0295520. doi: 10.1128/AEM.02955-20. Epub 2021 May 26. Appl Environ Microbiol. 2021. PMID: 33837012 Free PMC article.
-
Bacillaene, sharp objects consist in the arsenal of antibiotics produced by Bacillus.J Cell Physiol. 2024 Oct;239(10):e30974. doi: 10.1002/jcp.30974. Epub 2023 Feb 15. J Cell Physiol. 2024. PMID: 36790954 Review.
Cited by
-
Development of a Microbioreactor for Bacillus subtilis Biofilm Cultivation.Micromachines (Basel). 2024 Aug 15;15(8):1037. doi: 10.3390/mi15081037. Micromachines (Basel). 2024. PMID: 39203688 Free PMC article.
-
Complete genome sequence analysis and Pks genes identification of Brevibacillus brevis FJAT-0809-GLX with a broad inhibitory spectrum against phytopathogens.World J Microbiol Biotechnol. 2024 Oct 3;40(11):332. doi: 10.1007/s11274-024-04139-z. World J Microbiol Biotechnol. 2024. PMID: 39358614
-
Bacillus subtilis ensures high spore quality in competition with Salmonella Typhimurium via the SigB-dependent pathway.ISME J. 2025 Jan 2;19(1):wraf052. doi: 10.1093/ismejo/wraf052. ISME J. 2025. PMID: 40098255 Free PMC article.
-
Selective inhibition of the amyloid matrix of Escherichia coli biofilms by a bifunctional microbial metabolite.NPJ Biofilms Microbiomes. 2023 Oct 19;9(1):81. doi: 10.1038/s41522-023-00449-6. NPJ Biofilms Microbiomes. 2023. PMID: 37857690 Free PMC article.
-
Bacillus subtilis Intraspecies Interactions Shape Probiotic Activity Against Salmonella Typhimurium.Microb Biotechnol. 2024 Dec;17(12):e70065. doi: 10.1111/1751-7915.70065. Microb Biotechnol. 2024. PMID: 39718437 Free PMC article.
References
-
- Steenackers H, Hermans K, Vanderleyden J, De Keersmaecker SCJ. 2012. Salmonella biofilms: an overview on occurrence, structure, regulation and eradication. Food Res Int 45:502–531. doi:10.1016/j.foodres.2011.01.038. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

