Feasibility of home hand rehabilitation using musicglove after chronic spinal cord injury
- PMID: 36347833
- PMCID: PMC9643482
- DOI: 10.1038/s41394-022-00552-4
Feasibility of home hand rehabilitation using musicglove after chronic spinal cord injury
Abstract
Study design: Randomized, controlled single-blind cross over study. This study was registered on ClinicalTrials.gov (NCT02473614).
Objectives: Examine usership patterns and feasibility of MusicGlove for at home hand rehabilitation therapy following chronic spinal cord injury.
Setting: Homes of participants.
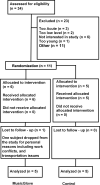
Methods: Ten participants with chronic spinal cord injury completed two baseline assessments of hand function. After a stable baseline was determined all participants were randomized into two groups: Experimental and Control. Each group was given a recommended therapy dosage. Following this participants switched interventions.
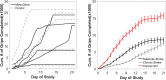
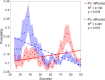
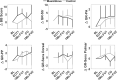
Results: On average participants had higher levels of compliance (6.1 ± 3.5 h.), and completed more grips (15,760 ± 9,590 grips) compared to participants in previous stroke studies using the same device. Participants modulated game parameters in a manner consistent with optimal challenge principles from motor learning theory. Participants in the experimental group increased their prehension ability (1 ± 1.4 MusicGlove, 0.2 ± 0.5 Control) and performance (1.4 ± 2.2 MusicGlove, 0.4 ± 0.55 Control) on the Graded and Redefined Assessment of Strength, Sensibility, and Prehension subtests. Increases in performance on the Box and Blocks Test also favored the experimental group compared to the conventional group at the end of therapy (4.2 ± 5.9, -1.0 ± 3.4 respectively).
Conclusions: MusicGlove is a feasible option for hand therapy in the home-setting for individuals with chronic SCI. Participants completed nearly twice as many gripping movements compared to individuals from the sub-acute and chronic stroke populations, and a number far greater than the number of movements typically achieved during traditional rehabilitation.
© 2022. The Author(s).
Conflict of interest statement
DJR has a financial interest in Hocoma A.G. and Flint Rehabilitation Devices LLC, companies that develop and sell rehabilitation devices. Flint sells the MusicGlove device examined in this study. The terms of these arrangements have been reviewed and approved by the University of California, Irvine, in accordance with its conflict of interest policies.
Figures

References
-
- Radomski MV. More than good intentions: Advancing adherence to therapy recommendations. The American journal of occupational therapy: official publication of the American Occupational Therapy Association. 2011;65:471–7. - PubMed
-
- Zondervan DK, Friedman N, Chang E, Zhao X, Augsburger R, Reinkensmeyer DJ, et al. Home-based hand rehabilitation after chronic stroke: Randomized, controlled single-blind trial comparing the MusicGlove with a conventional exercise program. J Rehabil Res Dev. 2016;53:457–72. doi: 10.1682/JRRD.2015.04.0057. - DOI - PubMed

