GDF11 promotes wound healing in diabetic mice via stimulating HIF-1ɑ-VEGF/SDF-1ɑ-mediated endothelial progenitor cell mobilization and neovascularization
- PMID: 36347996
- PMCID: PMC10104842
- DOI: 10.1038/s41401-022-01013-2
GDF11 promotes wound healing in diabetic mice via stimulating HIF-1ɑ-VEGF/SDF-1ɑ-mediated endothelial progenitor cell mobilization and neovascularization
Abstract
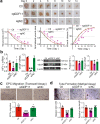
Non-healing diabetic wounds (DW) are a serious clinical problem that remained poorly understood. We recently found that topical application of growth differentiation factor 11 (GDF11) accelerated skin wound healing in both Type 1 DM (T1DM) and genetically engineered Type 2 diabetic db/db (T2DM) mice. In the present study, we elucidated the cellular and molecular mechanisms underlying the action of GDF11 on healing of small skin wound. Single round-shape full-thickness wound of 5-mm diameter with muscle and bone exposed was made on mouse dorsum using a sterile punch biopsy 7 days following the onset of DM. Recombinant human GDF11 (rGDF11, 50 ng/mL, 10 μL) was topically applied onto the wound area twice a day until epidermal closure (maximum 14 days). Digital images of wound were obtained once a day from D0 to D14 post-wounding. We showed that topical application of GDF11 accelerated the healing of full-thickness skin wounds in both type 1 and type 2 diabetic mice, even after GDF8 (a muscle growth factor) had been silenced. At the cellular level, GDF11 significantly facilitated neovascularization to enhance regeneration of skin tissues by stimulating mobilization, migration and homing of endothelial progenitor cells (EPCs) to the wounded area. At the molecular level, GDF11 greatly increased HIF-1ɑ expression to enhance the activities of VEGF and SDF-1ɑ, thereby neovascularization. We found that endogenous GDF11 level was robustly decreased in skin tissue of diabetic wounds. The specific antibody against GDF11 or silence of GDF11 by siRNA in healthy mice mimicked the non-healing property of diabetic wound. Thus, we demonstrate that GDF11 promotes diabetic wound healing via stimulating endothelial progenitor cells mobilization and neovascularization mediated by HIF-1ɑ-VEGF/SDF-1ɑ pathway. Our results support the potential of GDF11 as a therapeutic agent for non-healing DW.
Keywords: GDF11; HIF-1ɑ; VEGF/SDF-1α; diabetic wound; endothelial progenitor cell; neovascularization.
© 2022. The Author(s), under exclusive licence to Shanghai Institute of Materia Medica, Chinese Academy of Sciences and Chinese Pharmacological Society.
Conflict of interest statement
The authors declare no competing interests.
Figures






References
-
- Wukich DK, Raspovic KM, Suder NC. Patients with diabetic foot disease fear major lower-extremity amputation more than death. Foot Ankle Spec. 2017:1938640017694722. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

