Biomarkers of alveolar epithelial injury and endothelial dysfunction are associated with scores of pulmonary edema in invasively ventilated patients
- PMID: 36348302
- PMCID: PMC9799153
- DOI: 10.1152/ajplung.00185.2022
Biomarkers of alveolar epithelial injury and endothelial dysfunction are associated with scores of pulmonary edema in invasively ventilated patients
Abstract
Pulmonary edema is a central hallmark of acute respiratory distress syndrome (ARDS). Endothelial dysfunction and epithelial injury contribute to alveolar-capillary permeability but their differential contribution to pulmonary edema development remains understudied. Plasma levels of surfactant protein-D (SP-D), soluble receptor for advanced glycation end products (sRAGE), and angiopoietin-2 (Ang-2) were measured in a prospective, multicenter cohort of invasively ventilated patients. Pulmonary edema was quantified using the radiographic assessment of lung edema (RALE) and global lung ultrasound (LUS) score. Variables were collected within 48 h after intubation. Linear regression was used to examine the association of the biomarkers with pulmonary edema. In 362 patients, higher SP-D, sRAGE, and Ang-2 concentrations were significantly associated with higher RALE and global LUS scores. After stratification by ARDS subgroups (pulmonary, nonpulmonary, COVID, non-COVID), the positive association of SP-D levels with pulmonary edema remained, whereas sRAGE and Ang-2 showed less consistent associations throughout the subgroups. In a multivariable analysis, SP-D levels were most strongly associated with pulmonary edema when combined with sRAGE (RALE score: βSP-D = 6.79 units/log10 pg/mL, βsRAGE = 3.84 units/log10 pg/mL, R2 = 0.23; global LUS score: βSP-D = 3.28 units/log10 pg/mL, βsRAGE = 2.06 units/log10 pg/mL, R2 = 0.086), whereas Ang-2 did not further improve the model. Biomarkers of epithelial injury and endothelial dysfunction were associated with pulmonary edema in invasively ventilated patients. SP-D and sRAGE showed the strongest association, suggesting that epithelial injury may form a final common pathway in the alveolar-capillary barrier dysfunction underlying pulmonary edema.
Keywords: ARDS; endothelial dysfunction; epithelial injury; pulmonary edema; vascular permeability.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures

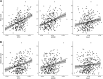
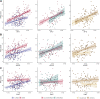

References
-
- van der Heijden M, Pickkers P, van Nieuw Amerongen GP, van Hinsbergh VWM, Bouw M, van der Hoeven JG, Groeneveld ABJ. Circulating angiopoietin-2 levels in the course of septic shock: relation with fluid balance, pulmonary dysfunction and mortality. Intensive Care Med 35: 1567–1574, 2009. doi:10.1007/s00134-009-1560-y. - DOI - PMC - PubMed
-
- Calfee CS, Delucchi K, Parsons PE, Thompson BT, Ware LB, Matthay MA; NHLBI ARDS Network. Subphenotypes in acute respiratory distress syndrome: latent class analysis of data from two randomised controlled trials. Lancet Respir Med 2: 611–620, 2014. doi:10.1016/S2213-2600(14)70097-9. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

