Tuft Cells: Context- and Tissue-Specific Programming for a Conserved Cell Lineage
- PMID: 36351364
- PMCID: PMC10443898
- DOI: 10.1146/annurev-pathol-042320-112212
Tuft Cells: Context- and Tissue-Specific Programming for a Conserved Cell Lineage
Abstract
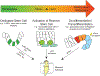
Tuft cells are found in tissues with distinct stem cell compartments, tissue architecture, and luminal exposures but converge on a shared transcriptional program, including expression of taste transduction signaling pathways. Here, we summarize seminal and recent findings on tuft cells, focusing on major categories of function-instigation of type 2 cytokine responses, orchestration of antimicrobial responses, and emerging roles in tissue repair-and describe tuft cell-derived molecules used to affect these functional programs. We review what is known about the development of tuft cells from epithelial progenitors under homeostatic conditions and during disease. Finally, we discuss evidence that immature, or nascent, tuft cells with potential for diverse functions are driven toward dominant effector programs by tissue- or perturbation-specific contextual cues, which may result in heterogeneous mature tuft cell phenotypes both within and between tissues.
Keywords: IL-25; acetylcholine; cysteinyl leukotriene; mucociliary clearance; tuft cell; type 2 immunity; type II taste transduction.
Figures


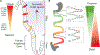
References
-
- von Moltke J 2018. Intestinal Tuft Cells. In Physiology of the Gastrointestinal Tract:721–33: Academic Press. Number of 721–33 pp.
-
- Bezencon C, Furholz A, Raymond F, Mansourian R, Metairon S, et al. 2008. Murine intestinal cells expressing Trpm5 are mostly brush cells and express markers of neuronal and inflammatory cells. J Comp Neurol 509:514–25 - PubMed

