Transcriptome and morphological analysis on the heart in gestational protein-restricted aging male rat offspring
- PMID: 36353510
- PMCID: PMC9638007
- DOI: 10.3389/fcell.2022.892322
Transcriptome and morphological analysis on the heart in gestational protein-restricted aging male rat offspring
Abstract
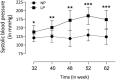
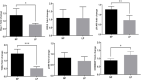
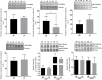
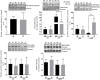
Background: Adverse factors that influence embryo/fetal development are correlated with increased risk of cardiovascular disease (CVD), type-2 diabetes, arterial hypertension, obesity, insulin resistance, impaired kidney development, psychiatric disorders, and enhanced susceptibility to oxidative stress and inflammatory processes in adulthood. Human and experimental studies have demonstrated a reciprocal relationship between birthweight and cardiovascular diseases, implying intrauterine adverse events in the onset of these abnormalities. In this way, it is plausible that confirmed functional and morphological heart changes caused by gestational protein restriction could be related to epigenetic effects anticipating cardiovascular disorders and reducing the survival time of these animals. Methods: Wistar rats were divided into two groups according to the protein diet content offered during the pregnancy: a normal protein diet (NP, 17%) or a Low-protein diet (LP, 6%). The arterial pressure was measured, and the cardiac mass, cardiomyocytes area, gene expression, collagen content, and immunostaining of proteins were performed in the cardiac tissue of male 62-weeks old NP compared to LP offspring. Results: In the current study, we showed a low birthweight followed by catch-up growth phenomena associated with high blood pressure development, increased heart collagen content, and cardiomyocyte area in 62-week-old LP offspring. mRNA sequencing analysis identified changes in the expression level of 137 genes, considering genes with a p-value < 0.05. No gene was. Significantly changed according to the adj-p-value. After gene-to-gene biological evaluation and relevance, the study demonstrated significant differences in genes linked to inflammatory activity, oxidative stress, apoptosis process, autophagy, hypertrophy, and fibrosis pathways resulting in heart function disorders. Conclusion: The present study suggests that gestational protein restriction leads to early cardiac diseases in the LP progeny. It is hypothesized that heart dysfunction is associated with fibrosis, myocyte hypertrophy, and multiple abnormal gene expression. Considering the above findings, it may suppose a close link between maternal protein restriction, specific gene expression, and progressive heart failure.
Keywords: arterial hypertension; cardiovascular disease; fetal programming; gestational protein-restriction; heart miRNA transcriptome; myocyte hypertrophy.
Copyright © 2022 Folguieri, Franco, Vieira, Gontijo and Boer.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




References
-
- Araújo E. P., De Souza C. T., Gasparetti A. L., Ueno M., Boschero A. C., Saad M. J. A., et al. (2005). Short-term in vivo inhibition of insulin receptor substrate-1 expression leads to insulin resistance, hyperinsulinemia, and increased adiposity. Endocrinology 146 (3), 1428–1437. 10.1210/en.2004-0778 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

