High-resolution spatiotemporal analysis of single serotonergic axons in an in vitro system
- PMID: 36353595
- PMCID: PMC9638127
- DOI: 10.3389/fnins.2022.994735
High-resolution spatiotemporal analysis of single serotonergic axons in an in vitro system
Abstract
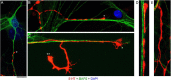
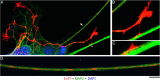
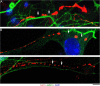
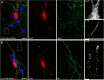
Vertebrate brains have a dual structure, composed of (i) axons that can be well-captured with graph-theoretical methods and (ii) axons that form a dense matrix in which neurons with precise connections operate. A core part of this matrix is formed by axons (fibers) that store and release 5-hydroxytryptamine (5-HT, serotonin), an ancient neurotransmitter that supports neuroplasticity and has profound implications for mental health. The self-organization of the serotonergic matrix is not well understood, despite recent advances in experimental and theoretical approaches. In particular, individual serotonergic axons produce highly stochastic trajectories, fundamental to the construction of regional fiber densities, but further advances in predictive computer simulations require more accurate experimental information. This study examined single serotonergic axons in culture systems (co-cultures and monolayers), by using a set of complementary high-resolution methods: confocal microscopy, holotomography (refractive index-based live imaging), and super-resolution (STED) microscopy. It shows that serotonergic axon walks in neural tissue may strongly reflect the stochastic geometry of this tissue and it also provides new insights into the morphology and branching properties of serotonergic axons. The proposed experimental platform can support next-generation analyses of the serotonergic matrix, including seamless integration with supercomputing approaches.
Keywords: 5-hydroxytryptamine (5-HT); axon; development; growth cone; in vitro; random walk; serotonergic; varicosities.
Copyright © 2022 Hingorani, Viviani, Sanfilippo and Janušonis.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

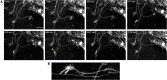




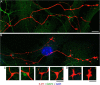

Similar articles
-
An experimental platform for stochastic analyses of single serotonergic fibers in the mouse brain.Front Neurosci. 2023 Oct 6;17:1241919. doi: 10.3389/fnins.2023.1241919. eCollection 2023. Front Neurosci. 2023. PMID: 37869509 Free PMC article.
-
Predicting the distribution of serotonergic axons: a supercomputing simulation of reflected fractional Brownian motion in a 3D-mouse brain model.Front Comput Neurosci. 2023 May 16;17:1189853. doi: 10.3389/fncom.2023.1189853. eCollection 2023. Front Comput Neurosci. 2023. PMID: 37265780 Free PMC article.
-
Serotonergic Axons as Fractional Brownian Motion Paths: Insights Into the Self-Organization of Regional Densities.Front Comput Neurosci. 2020 Jun 24;14:56. doi: 10.3389/fncom.2020.00056. eCollection 2020. Front Comput Neurosci. 2020. PMID: 32670042 Free PMC article.
-
The role of the serotonergic system in locomotor recovery after spinal cord injury.Front Neural Circuits. 2015 Feb 9;8:151. doi: 10.3389/fncir.2014.00151. eCollection 2014. Front Neural Circuits. 2015. PMID: 25709569 Free PMC article. Review.
-
Serotonergic mechanisms in spinal cord injury.Exp Neurol. 2019 Aug;318:174-191. doi: 10.1016/j.expneurol.2019.05.007. Epub 2019 May 11. Exp Neurol. 2019. PMID: 31085200 Review.
Cited by
-
An experimental platform for stochastic analyses of single serotonergic fibers in the mouse brain.Front Neurosci. 2023 Oct 6;17:1241919. doi: 10.3389/fnins.2023.1241919. eCollection 2023. Front Neurosci. 2023. PMID: 37869509 Free PMC article.
-
Predicting the distribution of serotonergic axons: a supercomputing simulation of reflected fractional Brownian motion in a 3D-mouse brain model.Front Comput Neurosci. 2023 May 16;17:1189853. doi: 10.3389/fncom.2023.1189853. eCollection 2023. Front Comput Neurosci. 2023. PMID: 37265780 Free PMC article.
-
The organization of serotonergic fibers in the Pacific angelshark brain: neuroanatomical and supercomputing analyses.Front Neurosci. 2025 Aug 8;19:1602116. doi: 10.3389/fnins.2025.1602116. eCollection 2025. Front Neurosci. 2025. PMID: 40860844 Free PMC article.
References
-
- Andersson M., Kjer H. M., Rafael-Patino J., Pacureanu A., Pakkenberg B., Thiran J. P., et al. (2020). Axon morphology is modulated by the local environment and impacts the noninvasive investigation of its structure-function relationship. Proc. Natl. Acad. Sci. U.S.A. 117 33649–33659. 10.1073/pnas.2012533117 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

