ACROBAT Edge: Safety and Efficacy of Switching Injected SRLs to Oral Paltusotine in Patients With Acromegaly
- PMID: 36353760
- PMCID: PMC10099171
- DOI: 10.1210/clinem/dgac643
ACROBAT Edge: Safety and Efficacy of Switching Injected SRLs to Oral Paltusotine in Patients With Acromegaly
Abstract
Context: Paltusotine is a once-daily, oral, nonpeptide small-molecule somatostatin receptor type 2 (SST2) agonist in clinical development for treatment of acromegaly.
Objective: This work aimed to evaluate change in insulin-like growth factor I (IGF-I) levels in patients switched from octreotide long-acting release or lanreotide depot monotherapy to paltusotine.
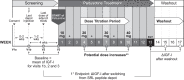
Methods: A phase 2, open-label, prospective, multicenter, multinational, nonrandomized, single-arm exploratory study was conducted in which dosage uptitrations were performed in a double-blinded manner. At 26 global sites, patients with acromegaly switched to paltusotine from injected somatostatin receptor ligand (SRL)-based therapy. Patients received 13-week treatment with once-daily oral paltusotine (10-40 mg/d). The primary end point was change from baseline to week 13 in IGF-I for patients who switched from long-acting octreotide or lanreotide depot monotherapy to paltusotine (group 1). All patients underwent a 4-week paltusotine washout at end of treatment period (wk 13-17). IGF-I, growth hormone (GH), patient-reported outcome, and safety data were collected.
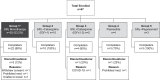
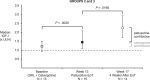
Results: Forty-seven patients enrolled. In group 1 (n = 25), IGF-I and GH showed no significant change between SRL baseline and end of paltusotine treatment at week 13 (median change in IGF-I = -0.03×upper limit of normal [ULN]; P = .6285; GH = -0.05 ng/mL; P = .6285). IGF-I and GH rose significantly in the 4 weeks after withdrawing paltusotine (median change in IGF-I = 0.55×ULN; P < .0001 [median increase 39%]; GH = 0.72 ng/mL; P < .0001 [109.1% increase]). No patients discontinued because of adverse events (AE); no treatment-related serious AEs were reported.
Conclusion: These results suggest once-daily oral paltusotine was effective in maintaining IGF-I values in patients with acromegaly who switched from injected SRLs. Paltusotine was well tolerated with a safety profile consistent with other SRLs.
Keywords: acromegaly; clinical trial; paltusotine; phase 2; somatostatin receptor ligands; somatostatin receptor type 2; somatotropinoma.
© The Author(s) 2022. Published by Oxford University Press on behalf of the Endocrine Society.
Figures



Comment in
-
Paltusotine, a Novel Oral Somatostatin Receptor Ligand in the Management of Acromegaly.J Clin Endocrinol Metab. 2023 Apr 13;108(5):e193-e194. doi: 10.1210/clinem/dgac762. J Clin Endocrinol Metab. 2023. PMID: 36582134 No abstract available.
References
-
- Colao A, Grasso LFS, Giustina A, et al. . Acromegaly. Nat Rev Dis Primers. 2019;5(1):20. - PubMed
-
- Colao A, Grasso LFS, Di Somma C, Pivonello R. Acromegaly and heart failure. Heart Fail Clin. 2019;15(3):399‐408. - PubMed
-
- Gadelha MR, Kasuki L, Lim DST, Fleseriu M. Systemic complications of acromegaly and the impact of the current treatment landscape: an update. Endocr Rev. 2019;40(1):268‐332. - PubMed
-
- Kasuki L, Antunes X, Lamback EB, Gadelha MR. Acromegaly: update on management and long-term morbidities. Endocrinol Metab Clin North Am. 2020;49(3):475‐486. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

