Palliative Treatment of Esophageal Cancer Using Calcium Electroporation
- PMID: 36358702
- PMCID: PMC9655404
- DOI: 10.3390/cancers14215283
Palliative Treatment of Esophageal Cancer Using Calcium Electroporation
Abstract
Calcium electroporation (CaEP) is a novel cancer therapy wherein high intracellular calcium levels, facilitated by reversible electroporation, trigger tumor necrosis. This study aimed to establish safety with CaEP within esophageal cancer. Patients with non-curable esophageal cancer were included at Copenhagen University Hospital Rigshospitalet in 2021 and 2022. In an outpatient setting, calcium gluconate was injected intratumorally followed by reversible electroporation applied with an endoscopic electrode. The primary endpoint was the prevalence of adverse events, followed by palliation of dysphagia. All patients were evaluated with CT and upper endoscopies up to two months after treatment. The trial was registered at ClinicalTrials.gov (NCT04958044). Eight patients were treated. One serious adverse event (anemia, requiring a single blood transfusion) and three adverse events (mild retrosternal pain (two) and oral thrush (one)) were registered. Initially, six patients suffered from dysphagia: two reported dysphagia relief and four reported no change. From the imaging evaluation, one patient had a partial response, three patients had no response, and four patients had progression. Six months after treatment, the patient who responded well was still in good condition and without the need for further oncological treatment. CaEP was conducted in eight patients with only a few side effects. This study opens the way for larger studies evaluating tumor regression and symptom palliation.
Keywords: calcium electroporation; esophageal cancer; malignant dysphagia; palliation.
Conflict of interest statement
Charlotte Egeland gave a presentation at an expert meeting hosted by Mirai Medical and Werfen in Barcelona on 21 April 2022. All expenses were covered by Mirai Medical. Experiences with the electrode and its usability was shared; results from the trial were not shared. Mirai Medical had no representatives in the investigator group, and they did not influence trial design, the performance of the trial, data processing, or manuscript preparation/reviewing. Besides this, the authors declare no conflict of interest.
Figures

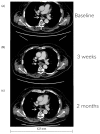
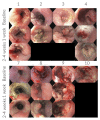
References
-
- American Cancer Society Survival Rates for Esophageal Cancer 2022. [(accessed on 9 June 2022)]. Available online: https://www.cancer.org/cancer/esophagus-cancer/detection-diagnosis-stagi....
-
- Kjaer D.W., Nassar M., Jensen L.S., Svendsen L.B., Mortensen F.V. A bridging stent to surgery in patients with esophageal and gastroesophageal junction cancer has a dramatic negative impact on patient survival: A retrospective cohort study through data acquired from a prospectively maintained national database. Dis. Esophagus. 2017;30:1–7. doi: 10.1111/dote.12474. - DOI - PubMed
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

