Submilligram Level of Beetle Antifreeze Proteins Minimize Cold-Induced Cell Swelling and Promote Cell Survival
- PMID: 36358934
- PMCID: PMC9687565
- DOI: 10.3390/biom12111584
Submilligram Level of Beetle Antifreeze Proteins Minimize Cold-Induced Cell Swelling and Promote Cell Survival
Abstract
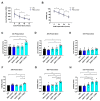
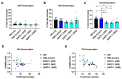
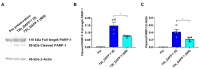
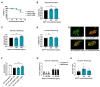
Hypothermic (cold) preservation is a limiting factor for successful cell and tissue transplantation where cell swelling (edema) usually develops, impairing cell function. University of Wisconsin (UW) solution, a standard cold preservation solution, contains effective components to suppress hypothermia-induced cell swelling. Antifreeze proteins (AFPs) found in many cold-adapted organisms can prevent cold injury of the organisms. Here, the effects of a beetle AFP from Dendroides canadensis (DAFP-1) on pancreatic β-cells preservation were first investigated. As low as 500 µg/mL, DAFP-1 significantly minimized INS-1 cell swelling and subsequent cell death during 4 °C preservation in UW solution for up to three days. However, such significant cytoprotection was not observed by an AFP from Tenebrio molitor (TmAFP), a structural homologue to DAFP-1 but lacking arginine, at the same levels. The cytoprotective effect of DAFP-1 was further validated with the primary β-cells in the isolated rat pancreatic islets in UW solution. The submilligram level supplement of DAFP-1 to UW solution significantly increased the islet mass recovery after three days of cold preservation followed by rewarming. The protective effects of DAFP-1 in UW solution were discussed at a molecular level. The results indicate the potential of DAFP-1 to enhance cell survival during extended cold preservation.
Keywords: antifreeze protein; cell swelling; post-hypothermic preservation survival.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
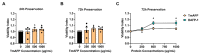
Similar articles
-
Interaction of reduced nicotinamide adenine dinucleotide with an antifreeze protein from Dendroides canadensis: mechanistic implication of antifreeze activity enhancement.J Mol Recognit. 2011 Nov-Dec;24(6):1025-32. doi: 10.1002/jmr.1151. J Mol Recognit. 2011. PMID: 22038809 Free PMC article.
-
Arginine, a key residue for the enhancing ability of an antifreeze protein of the beetle Dendroides canadensis.Biochemistry. 2009 Oct 13;48(40):9696-703. doi: 10.1021/bi901283p. Biochemistry. 2009. PMID: 19746966 Free PMC article.
-
Toxicity profiles and protective effects of antifreeze proteins from insect in mammalian models.Toxicol Lett. 2022 Sep 1;368:9-23. doi: 10.1016/j.toxlet.2022.07.009. Epub 2022 Jul 25. Toxicol Lett. 2022. PMID: 35901986 Free PMC article.
-
Recombinant Dendroides canadensis antifreeze proteins as potential ingredients in cryopreservation solutions.Cryobiology. 2014 Jun;68(3):411-8. doi: 10.1016/j.cryobiol.2014.03.006. Epub 2014 Mar 21. Cryobiology. 2014. PMID: 24662031 Free PMC article.
-
Efficiency of beetle (Dendroides canadensis) recombinant antifreeze protein for buffalo semen freezability and fertility.Theriogenology. 2016 Oct 15;86(7):1662-9. doi: 10.1016/j.theriogenology.2016.05.028. Epub 2016 May 30. Theriogenology. 2016. PMID: 27349137
References
-
- Bhatnagar B., Zakharov B., Fisyuk A., Wen X., Karim F., Lee K., Seryotkin Y., Mogodi M., Fitch A., Boldyreva E., et al. Protein/Ice Interaction: High-Resolution Synchrotron X-ray Diffraction Differentiates Pharmaceutical Proteins from Lysozyme. J. Phys. Chem. B. 2019;123:5690–5699. doi: 10.1021/acs.jpcb.9b02443. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

