A New Model of Acute Exacerbation of Experimental Pulmonary Fibrosis in Mice
- PMID: 36359778
- PMCID: PMC9654438
- DOI: 10.3390/cells11213379
A New Model of Acute Exacerbation of Experimental Pulmonary Fibrosis in Mice
Abstract
Rationale: idiopathic pulmonary fibrosis (IPF) is the most severe form of fibrosing interstitial lung disease, characterized by progressive respiratory failure leading to death. IPF's natural history is heterogeneous, and its progression unpredictable. Most patients develop a progressive decline of respiratory function over years; some remain stable, but others present a fast-respiratory deterioration without identifiable cause, classified as acute exacerbation (AE).
Objectives: to develop and characterize an experimental mice model of lung fibrosis AE, mimicking IPF-AE at the functional, histopathological, cellular and molecular levels.
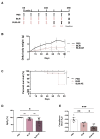
Methods: we established in C57BL/6 male mice a chronic pulmonary fibrosis using a repetitive low-dose bleomycin (BLM) intratracheal (IT) instillation regimen (four instillations of BLM every 2 weeks), followed by two IT instillations of a simple or double-dose BLM challenge to induce AE. Clinical follow-up and histological and molecular analyses were done for fibrotic and inflammatory lung remodeling analysis.
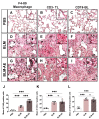
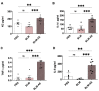
Measurements and main results: as compared with a low-dose BLM regimen, this AE model induced a late burst of animal mortality, worsened lung fibrosis and remodeling, and superadded histopathological features as observed in humans IPF-AE. This was associated with stronger inflammation, increased macrophage infiltration of lung tissue and increased levels of pro-inflammatory cytokines in lung homogenates. Finally, it induced in the remodeled lung a diffuse expression of hypoxia-inducible factor 1α, a hallmark of tissular hypoxia response and a major player in the progression of IPF.
Conclusion: this new model is a promising model of AE in chronic pulmonary fibrosis that could be relevant to mimic IPF-AE in preclinical trials.
Keywords: acute exacerbation; animal model; inflammation; lung fibrosis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Cottin V., Spagnolo P., Bonniaud P., Nolin M., Dalon F., Kirchgässler K.U., Kamath T.V., Van Ganse E., Belhassen M. Mortality and Respiratory-Related Hospitalizations in Idiopathic Pulmonary Fibrosis Not Treated With Antifibrotics. Front. Med. 2021;8:802989. doi: 10.3389/fmed.2021.802989. - DOI - PMC - PubMed
-
- Collard H.R., Ryerson C.J., Corte T.J., Jenkins G., Kondoh Y., Lederer D.J., Lee J.S., Maher T.M., Wells A.U., Antoniou K.M., et al. Acute Exacerbation of Idiopathic Pulmonary Fibrosis. An International Working Group Report. Am. J. Respir. Crit. Care Med. 2016;194:265–275. doi: 10.1164/rccm.201604-0801CI. - DOI - PubMed
-
- Maher T.M., Whyte M.K.B., Hoyles R.K., Parfrey H., Ochiai Y., Mathieson N., Turnbull A., Williamson N., Bennett B.M. Development of a Consensus Statement for the Definition, Diagnosis, and Treatment of Acute Exacerbations of Idiopathic Pulmonary Fibrosis Using the Delphi Technique. Adv. Ther. 2015;32:929–943. doi: 10.1007/s12325-015-0249-6. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

