Necroptosis: A Pathogenic Negotiator in Human Diseases
- PMID: 36361505
- PMCID: PMC9655262
- DOI: 10.3390/ijms232112714
Necroptosis: A Pathogenic Negotiator in Human Diseases
Abstract
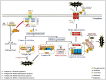
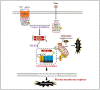
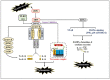
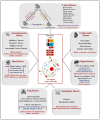
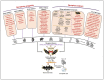
Over the past few decades, mechanisms of programmed cell death have attracted the scientific community because they are involved in diverse human diseases. Initially, apoptosis was considered as a crucial mechanistic pathway for programmed cell death; recently, an alternative regulated mode of cell death was identified, mimicking the features of both apoptosis and necrosis. Several lines of evidence have revealed that dysregulation of necroptosis leads to pathological diseases such as cancer, cardiovascular, lung, renal, hepatic, neurodegenerative, and inflammatory diseases. Regulated forms of necrosis are executed by death receptor ligands through the activation of receptor-interacting protein kinase (RIPK)-1/3 and mixed-lineage kinase domain-like (MLKL), resulting in the formation of a necrosome complex. Many papers based on genetic and pharmacological studies have shown that RIPKs and MLKL are the key regulatory effectors during the progression of multiple pathological diseases. This review focused on illuminating the mechanisms underlying necroptosis, the functions of necroptosis-associated proteins, and their influences on disease progression. We also discuss numerous natural and chemical compounds and novel targeted therapies that elicit beneficial roles of necroptotic cell death in malignant cells to bypass apoptosis and drug resistance and to provide suggestions for further research in this field.
Keywords: MLKL; RIPK1; RIPK3; apoptosis; human diseases; necroptosis.
Conflict of interest statement
The authors declare that there are no conflict of interests.
Figures
References
-
- Galluzzi L., Vitale I., Abrams J.M., Alnemri E.S., Baehrecke E.H., Blagosklonny M.V., Dawson T.M., Dawson V.L., El-Deiry W.S., Fulda S., et al. Molecular definitions of cell death subroutines: Recommendations of the Nomenclature Committee on Cell Death 2012. Cell Death Differ. 2012;19:107–120. doi: 10.1038/cdd.2011.96. - DOI - PMC - PubMed
-
- Ge Yan M.E. Thomas Efferth, Multiple cell death modalities and their key features. World Acad. Sci. 2020;2:39–48.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

