Biophysical Characterization of Adeno-Associated Virus Vectors Using Ion-Exchange Chromatography Coupled to Light Scattering Detectors
- PMID: 36361506
- PMCID: PMC9655919
- DOI: 10.3390/ijms232112715
Biophysical Characterization of Adeno-Associated Virus Vectors Using Ion-Exchange Chromatography Coupled to Light Scattering Detectors
Abstract
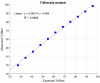
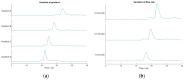
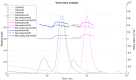
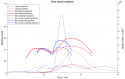
Ion-exchange chromatography coupled to light scattering detectors represents a fast and simple analytical method for the assessment of multiple critical quality attributes (CQA) in one single measurement. The determination of CQAs play a crucial role in Adeno-Associated Virus (AAV)-based gene therapies and their applications in humans. Today, several different analytical techniques, including size-exclusion chromatography (SEC), analytical ultracentrifugation (AUC), qPCR or ELISA, are commonly used to characterize the gene therapy product regarding capsid titer, packaging efficiency, vector genome integrity, aggregation content and other process-related impurities. However, no universal method for the simultaneous determination of multiple CQAs is currently available. Here, we present a novel robust ion-exchange chromatography method coupled to multi-angle light scattering detectors (IEC-MALS) for the comprehensive characterization of empty and filled AAVs concerning capsid titer, full-to-total ratio, absolute molar mass of the protein and nucleic acid, and the size and polydispersity without baseline-separation of both species prior to data analysis. We demonstrate that the developed IEC-MALS assay is applicable to different serotypes and can be used as an orthogonal method to other established analytical techniques.
Keywords: adeno-associated virus vectors; critical quality attributes; dynamic light scattering; ion-exchange chromatography; multi-angle light scattering; protein characterization.
Conflict of interest statement
Authors Christina Wagner, Martin Lemmerer, Bernd Innthaler and Robert Pletzenauer are current full-time employees of Baxalta Innovations GmbH and own company stocks. Ruth Birner-Gruenberger declares no conflict of interest.
Figures




References
-
- Wang C., Mulagapati S.H.R., Chen Z., Du J., Zhao X., Xi G., Chen L., Linke T., Gao C., Schmelzer A.E., et al. Developing an Anion Exchange Chromatography Assay for Determining Empty and Full Capsid Contents in AAV6.2. Mol. Ther.—Methods Clin. Dev. 2019;15:257–263. doi: 10.1016/j.omtm.2019.09.006. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

