Sustained Activation of CLR/RAMP Receptors by Gel-Forming Agonists
- PMID: 36362188
- PMCID: PMC9655119
- DOI: 10.3390/ijms232113408
Sustained Activation of CLR/RAMP Receptors by Gel-Forming Agonists
Abstract
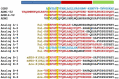
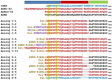
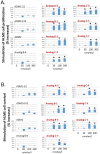
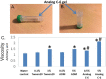
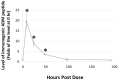
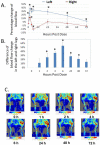
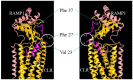
Background: Adrenomedullin (ADM), adrenomedullin 2 (ADM2), and CGRP family peptides are important regulators of vascular vasotone and integrity, neurotransmission, and fetoplacental development. These peptides signal through CLR/RAMP1, 2, and 3 receptors, and protect against endothelial dysfunction in disease models. As such, CLR/RAMP receptor agonists are considered important therapeutic candidates for various diseases. Methods and Results: Based on the screening of a series of palmitoylated chimeric ADM/ADM2 analogs, we demonstrated a combination of lipidation and accommodating motifs at the hinge region of select peptides is important for gaining an enhanced receptor-activation activity and improved stimulatory effects on the proliferation and survival of human lymphatic endothelial cells when compared to wild-type peptides. In addition, by serendipity, we found that select palmitoylated analogs self-assemble to form liquid gels, and subcutaneous administration of an analog gel led to the sustained presence of the peptide in the circulation for >2 days. Consistently, subcutaneous injection of the analog gel significantly reduced the blood pressure in SHR rats and increased vasodilation in the hindlimbs of adult rats for days. Conclusions: Together, these data suggest gel-forming adrenomedullin analogs may represent promising candidates for the treatment of various life-threatening endothelial dysfunction-associated diseases such as treatment-resistant hypertension and preeclampsia, which are in urgent need of an effective drug.
Keywords: CLR/RAMP receptor; RAMP2; adrenomedullin; endothelium; liquid gel; treatment-resistant hypertension; vasodilation.
Conflict of interest statement
The author (S.Y.T.H.) has pending patent applications (U.S. Application Serial No. 62643593, Gel-forming polypeptides) regarding the gel-forming analogs described here. Author SYTH is employed by Adepthera LLC. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures


Similar articles
-
Gel-forming antagonist provides a lasting effect on CGRP-induced vasodilation.Front Pharmacol. 2022 Dec 8;13:1040951. doi: 10.3389/fphar.2022.1040951. eCollection 2022. Front Pharmacol. 2022. PMID: 36569288 Free PMC article.
-
Development of chimeric and bifunctional antagonists for CLR/RAMP receptors.PLoS One. 2019 May 31;14(5):e0216996. doi: 10.1371/journal.pone.0216996. eCollection 2019. PLoS One. 2019. PMID: 31150417 Free PMC article.
-
Calcitonin Gene Related Peptide, Adrenomedullin, and Adrenomedullin 2 Function in Uterine Artery During Human Pregnancy.Endocrinology. 2022 Jan 1;163(1):bqab204. doi: 10.1210/endocr/bqab204. Endocrinology. 2022. PMID: 34558598 Free PMC article.
-
Regulation of cardiovascular development and homeostasis by the adrenomedullin-RAMP system.Peptides. 2019 Jan;111:55-61. doi: 10.1016/j.peptides.2018.04.004. Epub 2018 Apr 22. Peptides. 2019. PMID: 29689347 Review.
-
Roles of CLR/RAMP receptor signaling in reproduction and development.Curr Protein Pept Sci. 2013 Aug;14(5):393-406. doi: 10.2174/13892037113149990056. Curr Protein Pept Sci. 2013. PMID: 23745703 Free PMC article. Review.
Cited by
-
Gel-forming antagonist provides a lasting effect on CGRP-induced vasodilation.Front Pharmacol. 2022 Dec 8;13:1040951. doi: 10.3389/fphar.2022.1040951. eCollection 2022. Front Pharmacol. 2022. PMID: 36569288 Free PMC article.
-
Clinical Potential of Adrenomedullin Signaling in the Cardiovascular System.Circ Res. 2023 Apr 28;132(9):1185-1202. doi: 10.1161/CIRCRESAHA.123.321673. Epub 2023 Apr 27. Circ Res. 2023. PMID: 37104556 Free PMC article. Review.
-
Novel Calcitonin Gene-Related Peptide (CGRP) Interfering Migraine Therapies and Stroke-A Review.Int J Mol Sci. 2024 Oct 30;25(21):11685. doi: 10.3390/ijms252111685. Int J Mol Sci. 2024. PMID: 39519240 Free PMC article. Review.
-
CGRP-Targeted Migraine Therapies in Patients With Vascular Risk Factors or Stroke: A Review.Neurology. 2025 Jul 22;105(2):e213852. doi: 10.1212/WNL.0000000000213852. Epub 2025 Jun 19. Neurology. 2025. PMID: 40537071 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

