Sources, Transformations, Syntheses, and Bioactivities of Monoterpene Pyridine Alkaloids and Cyclopenta[c]pyridine Derivatives
- PMID: 36364013
- PMCID: PMC9656638
- DOI: 10.3390/molecules27217187
Sources, Transformations, Syntheses, and Bioactivities of Monoterpene Pyridine Alkaloids and Cyclopenta[c]pyridine Derivatives
Abstract
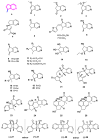
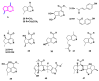
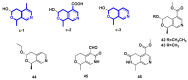
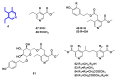
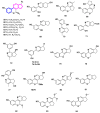
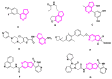
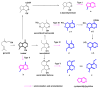
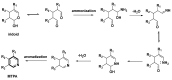
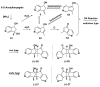
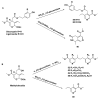
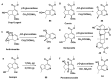
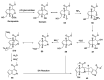
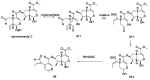
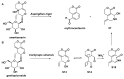
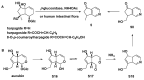
Monoterpene pyridine alkaloids (MTPAs) are alkaloids derived from iridoid glycosides (IGs). The common molecular structure of MTPAs is the pyridine ring, while some of them have a cyclopenta[c]pyridine skeleton. Some compounds containing this structure are potentially bioactive medicinal agents. In this paper, seven drug candidates (A-G), ninety natural source products (1-90), thirty-seven synthesized compounds (91-127), as well as twenty-six key intermediates (S1-S26) were summarized. We categorized five types of MTPAs and one type of cyclopenta[c]pyridine alkaloids in all. Additionally, their possible genetic pathways were proposed. Then, the chemical transformation, biotransformation, chemical synthesis, as well as the bioactivity of MTPAs and cyclopenta[c]pyridine derivatives were analyzed and summarized. Cyclopenta[c]pyridine derivatives can be concisely and chirally synthesized, and they have shown potentials with antibacterial, insecticidal, antiviral, anti-inflammatory, and neuropharmacological activities.
Keywords: bioactivity; cyclopenta[c]pyridine derivatives; monoterpene pyridine alkaloids; source; synthesis; transformation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
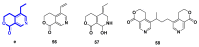
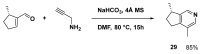
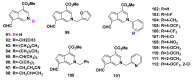
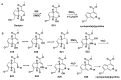
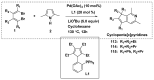
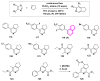
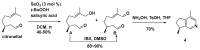
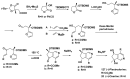
References
-
- Dyachenko I.V., Dyachenko V.D. Cycloalka [c] pyridine derivatives. Methods of synthesis and chemical properties. Russ. J. Org. Chem. 2017;53:1769–1787. doi: 10.1134/S1070428017120016. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

