Methods of Lysergic Acid Synthesis-The Key Ergot Alkaloid
- PMID: 36364148
- PMCID: PMC9654825
- DOI: 10.3390/molecules27217322
Methods of Lysergic Acid Synthesis-The Key Ergot Alkaloid
Abstract
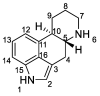
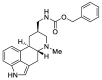
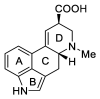
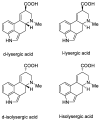
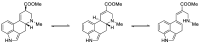
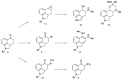
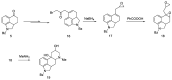
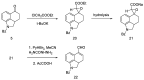
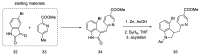
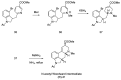
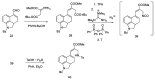
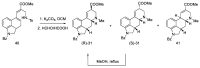
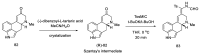
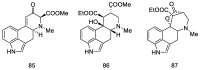
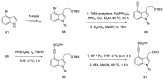
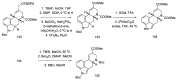
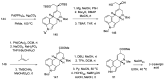
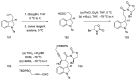
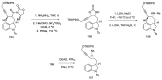
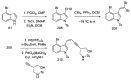
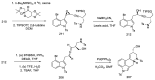
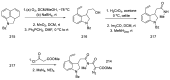
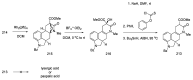
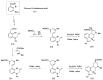
Ergot is the spore form of the fungus Claviceps purpurea. Ergot alkaloids are indole compounds that are biosynthetically derived from L-tryptophan and represent the largest group of fungal nitrogen metabolites found in nature. The common part of ergot alkaloids is lysergic acid. This review shows the importance of lysergic acid as a representative of ergot alkaloids. The subject of ergot and its alkaloids is presented, with a particular focus on lysergic acid. All methods of total lysergic acid synthesis-through Woodward, Hendrickson, and Szantay intermediates and Heck coupling methods-are presented. The topic of biosynthesis is also discussed.
Keywords: biosynthesis; ergot alkaloids; lysergic acid; total synthesis.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures
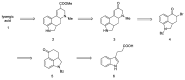
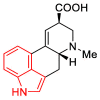
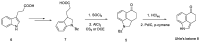
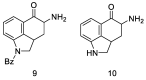
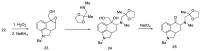
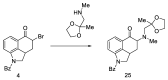
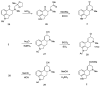
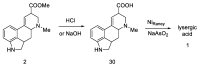
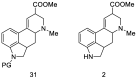
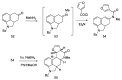
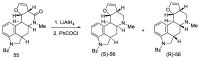
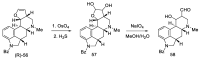
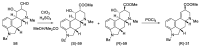
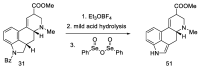
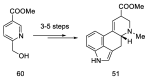
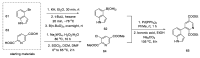
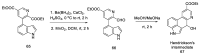
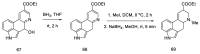
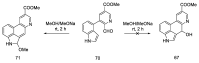
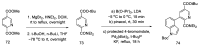
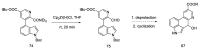
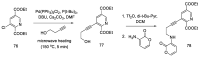
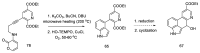
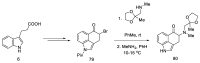
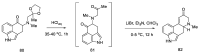
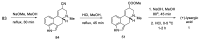
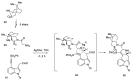
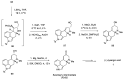
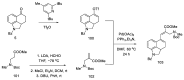
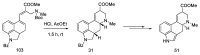
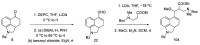
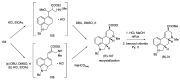

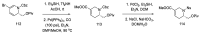
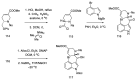
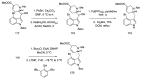
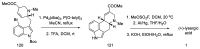
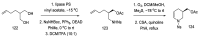
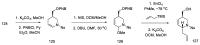
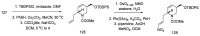
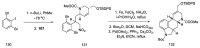
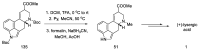
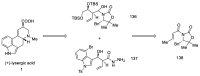
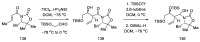
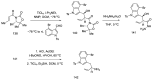
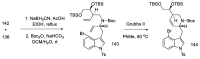
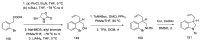
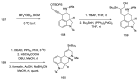
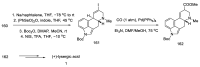
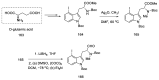
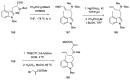
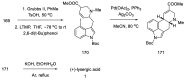
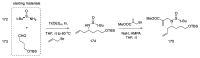
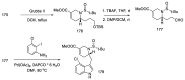
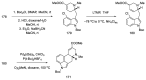

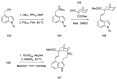
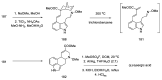
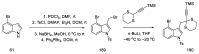
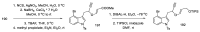
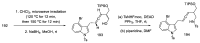
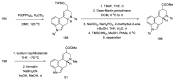

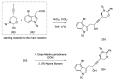
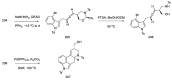

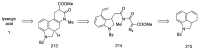
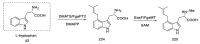

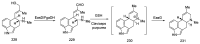
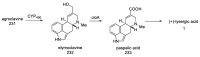
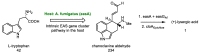
References
-
- Ochodzki P., National Research Institute . Zapobieganie Zagrożeniom Związanym z Występowaniem w Ziarnie Zbóż Alkaloidów Pasożytniczego Grzyba Buławinki Czerwonej (Sporysz)—Prevention of Risks Associated with the Presence of Alkaloids in the Cereal Grain Parasitic Fungus of Ergot. Expertise; Radzików, Poland: 2015.
-
- Berde B., Schild H.O., editors. Ergot Alkaloids and Related Compounds. Springer; Berlin/Heidelberg, Germany: 1978.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

