Increase of mast cells in COVID-19 pneumonia may contribute to pulmonary fibrosis and thrombosis
- PMID: 36366933
- PMCID: PMC9877713
- DOI: 10.1111/his.14838
Increase of mast cells in COVID-19 pneumonia may contribute to pulmonary fibrosis and thrombosis
Abstract
Aims: Lung tissue from COVID-19 patients shares similar histomorphological features with chronic lung allograft disease, also suggesting activation of autoimmune-related pathways in COVID-19. To more clearly understand the underlying spectrum of pathophysiology in COVID-19 pneumonia, we analysed mRNA expression of autoimmune-related genes in post-mortem lung tissue from COVID-19 patients.
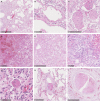
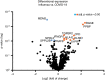
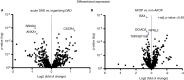
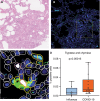
Methods and results: Formalin-fixed, paraffin-embedded lung tissue samples of 18 COVID-19 patients and eight influenza patients were used for targeted gene expression profiling using NanoString technology. Multiplex immunofluorescence for tryptase and chymase was applied for validation. Genes related to mast cells were significantly increased in COVID-19. This finding was strengthened by multiplex immunofluorescence also showing a significant increase of tryptase- and chymase-positive cells in COVID-19. Furthermore, receptors for advanced glycation end-products (RAGE) and pro-platelet basic protein (PPBP) were up-regulated in COVID-19 compared to influenza. Genes associated with Type I interferon signalling showed a significant correlation to detected SARS-CoV2 pathway-related genes. The comparison of lung tissue samples from both groups based on the presence of histomorphological features indicative of acute respiratory distress syndrome did not result in finding any specific gene or pathways.
Conclusion: Two separate means of measuring show a significant increase of mast cells in SARS-CoV-2-infected lung tissue compared to influenza. Additionally, several genes involved in fibrosis and thrombosis, among which are RAGE and PPBP, are up-regulated in COVID-19. As mast cells are able to induce thrombosis and fibrosis, they may play an important role in the pathogenesis of COVID-19.
Keywords: COVID-19; autoimmunity; fibrosis; gene expression profiling; genomics; mast cells; thrombosis.
© 2022 The Authors. Histopathology published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare that they have no conflicts of interest and no source of support to declare.
Figures

Similar articles
-
Role of mast cells in the pathogenesis of severe lung damage in COVID-19 patients.Respir Res. 2022 Dec 21;23(1):371. doi: 10.1186/s12931-022-02284-3. Respir Res. 2022. PMID: 36544127 Free PMC article.
-
Increase in venous thromboembolism in SARS-CoV-2 infected lung tissue: proteome analysis of lung parenchyma, isolated endothelium, and thrombi.Histopathology. 2024 May;84(6):967-982. doi: 10.1111/his.15143. Epub 2024 Jan 22. Histopathology. 2024. PMID: 38253958
-
Severity of SARS-CoV-2 infection is associated with high numbers of alveolar mast cells and their degranulation.Front Immunol. 2022 Sep 26;13:968981. doi: 10.3389/fimmu.2022.968981. eCollection 2022. Front Immunol. 2022. PMID: 36225927 Free PMC article.
-
Molecular Pathogenesis of Fibrosis, Thrombosis and Surfactant Dysfunction in the Lungs of Severe COVID-19 Patients.Biomolecules. 2022 Dec 10;12(12):1845. doi: 10.3390/biom12121845. Biomolecules. 2022. PMID: 36551272 Free PMC article. Review.
-
Tryptase immunoreactive mast cell hyperplasia in bronchopulmonary dysplasia.Pediatr Pulmonol. 1995 Jun;19(6):336-43. doi: 10.1002/ppul.1950190605. Pediatr Pulmonol. 1995. PMID: 7567212 Review.
Cited by
-
Recombinant SARS-CoV-2 Spike Protein Stimulates Secretion of Chymase, Tryptase, and IL-1β from Human Mast Cells, Augmented by IL-33.Int J Mol Sci. 2023 May 30;24(11):9487. doi: 10.3390/ijms24119487. Int J Mol Sci. 2023. PMID: 37298438 Free PMC article.
-
The plasma proteome differentiates the multisystem inflammatory syndrome in children (MIS-C) from children with SARS-CoV-2 negative sepsis.Mol Med. 2024 Apr 17;30(1):51. doi: 10.1186/s10020-024-00806-x. Mol Med. 2024. PMID: 38632526 Free PMC article.
-
Dysregulated early transcriptional signatures linked to mast cell and interferon responses are implicated in COVID-19 severity.Front Immunol. 2023 May 16;14:1166574. doi: 10.3389/fimmu.2023.1166574. eCollection 2023. Front Immunol. 2023. PMID: 37261339 Free PMC article.
-
Transfusion-Related Acute Lung Injury: from Mechanistic Insights to Therapeutic Strategies.Adv Sci (Weinh). 2025 Mar;12(11):e2413364. doi: 10.1002/advs.202413364. Epub 2025 Jan 21. Adv Sci (Weinh). 2025. PMID: 39836498 Free PMC article. Review.
-
Proteomic signatures of vaccine-induced and breakthrough infection-induced host responses to SARS-CoV-2.Vaccine. 2025 Jan 1;43(Pt 1):126484. doi: 10.1016/j.vaccine.2024.126484. Epub 2024 Nov 8. Vaccine. 2025. PMID: 39520894
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

