Phase II Randomized Study of Salvage Radiation Therapy Plus Enzalutamide or Placebo for High-Risk Prostate-Specific Antigen Recurrent Prostate Cancer After Radical Prostatectomy: The SALV-ENZA Trial
- PMID: 36367998
- PMCID: PMC9940936
- DOI: 10.1200/JCO.22.01662
Phase II Randomized Study of Salvage Radiation Therapy Plus Enzalutamide or Placebo for High-Risk Prostate-Specific Antigen Recurrent Prostate Cancer After Radical Prostatectomy: The SALV-ENZA Trial
Abstract
Purpose: We sought to investigate whether enzalutamide (ENZA), without concurrent androgen deprivation therapy, increases freedom from prostate-specific antigen (PSA) progression (FFPP) when combined with salvage radiation therapy (SRT) in men with recurrent prostate cancer after radical prostatectomy (RP).
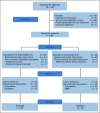
Patients and methods: Men with biochemically recurrent prostate cancer after RP were enrolled into a randomized, double-blind, phase II, placebo-controlled, multicenter study of SRT plus ENZA or placebo (ClinicalTrials.gov identifier: NCT02203695). Random assignment (1:1) was stratified by center, surgical margin status (R0 v R1), PSA before salvage treatment (PSA ≥ 0.5 v < 0.5 ng/mL), and pathologic Gleason sum (7 v 8-10). Patients were assigned to receive either ENZA 160 mg once daily or matching placebo for 6 months. After 2 months of study drug therapy, external-beam radiation (66.6-70.2 Gy) was administered to the prostate bed (no pelvic nodes). The primary end point was FFPP in the intention-to-treat population. Secondary end points were time to local recurrence within the radiation field, metastasis-free survival, and safety as determined by frequency and severity of adverse events.
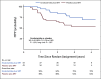
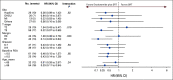
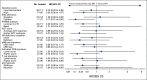
Results: Eighty-six (86) patients were randomly assigned, with a median follow-up of 34 (range, 0-52) months. Trial arms were well balanced. The median pre-SRT PSA was 0.3 (range, 0.06-4.6) ng/mL, 56 of 86 patients (65%) had extraprostatic disease (pT3), 39 of 86 (45%) had a Gleason sum of 8-10, and 43 of 86 (50%) had positive surgical margins (R1). FFPP was significantly improved with ENZA versus placebo (hazard ratio [HR], 0.42; 95% CI, 0.19 to 0.92; P = .031), and 2-year FFPP was 84% versus 66%, respectively. Subgroup analyses demonstrated differential benefit of ENZA in men with pT3 (HR, 0.22; 95% CI, 0.07 to 0.69) versus pT2 disease (HR, 1.54; 95% CI, 0.43 to 5.47; Pinteraction = .019) and R1 (HR, 0.14; 95% CI, 0.03 to 0.64) versus R0 disease (HR, 1.00; 95% CI, 0.36 to 2.76; Pinteraction = .023). There were insufficient secondary end point events for analysis. The most common adverse events were grade 1-2 fatigue (65% ENZA v 53% placebo) and urinary frequency (40% ENZA v 49% placebo).
Conclusion: SRT plus ENZA monotherapy for 6 months in men with PSA-recurrent high-risk prostate cancer after RP is safe and delays PSA progression relative to SRT alone. The impact of ENZA on distant metastasis or survival is unknown at this time.
Conflict of interest statement
Phase II Randomized Study of Salvage Radiation Therapy Plus Enzalutamide or Placebo for High-Risk Prostate-Specific Antigen Recurrent Prostate Cancer After Radical Prostatectomy: The SALV-ENZA Trial
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated unless otherwise noted. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to
Open Payments is a public database containing information reported by companies about payments made to US-licensed physicians (
Figures
References
-
- Siegel RL, Miller KD, Fuchs HE, Jemal A: Cancer statistics, 2022. CA Cancer J Clin 72:7-33, 2022 - PubMed
-
- Spratt DE, Dess RT, Zumsteg ZS, et al. : A systematic review and framework for the use of hormone therapy with salvage radiation therapy for recurrent prostate cancer. Eur Urol 73:156-165, 2018 - PubMed
-
- Cornford P, van den Bergh RCN, Briers E, et al. : EAU-EANM-ESTRO-ESUR-SIOG guidelines on prostate cancer. Part II-2020 update: Treatment of relapsing and metastatic prostate cancer. Eur Urol 79:263-282, 2021 - PubMed
-
- Schaeffer E, Srinivas S, Antonarakis ES, et al. : NCCN guidelines insights: Prostate cancer, version 1.2021. J Natl Compr Canc Netw 19:134-143, 2021 - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

