Emergent Organoboron Acid Catalysts
- PMID: 36374612
- PMCID: PMC10183059
- DOI: 10.1021/acs.joc.2c01695
Emergent Organoboron Acid Catalysts
Abstract
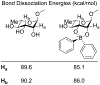
Organoboron acids are stable, organic-soluble Lewis acids with potential application as catalysts for a wide variety of chemical reactions. In this review, we summarize the utility of boronic and borinic acids, as well as boric acid, as catalysts for organic transformations. Typically, the catalytic processes exploit the Lewis acidity of trivalent boron, enabling the reversible formation of a covalent bond with oxygen. Our focus is on recent developments in the catalysis of dehydration, carbonyl condensation, acylation, alkylation, and cycloaddition reactions. We conclude that organoboron acids have a highly favorable prospectus as the source of new catalysts.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
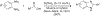
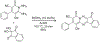

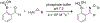
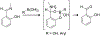
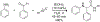
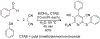
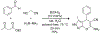
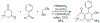

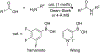
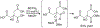
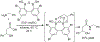
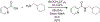
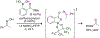
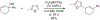
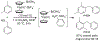
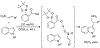
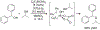
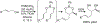
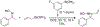
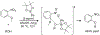

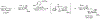

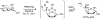
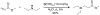
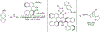

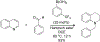
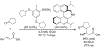
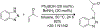
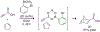
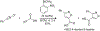
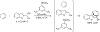
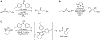
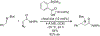
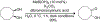

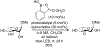
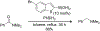
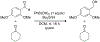
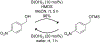
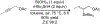
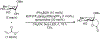
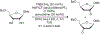
References
-
- Suzuki A Cross-coupling reactions of organoboranes: An easy way to construct C–C bonds (Nobel lecture). Angew. Chem., Int. Ed 2011, 50, 6722–6737. - PubMed
-
- Brown DG; Boström J Analysis of past and present synthetic methodologies on medicinal chemistry: Where have all the new reactions gone? J. Med. Chem 2016, 59, 4443–4458. - PubMed
-
- Beletskaya IP; Alonso F; Tyurin V The Suzuki–Miyaura reaction after the Nobel prize. Coord. Chem. Rev 2019, 385, 137–173.
-
- Fernández E; Whiting A, Eds. Synthesis and Application of Organoboron Compounds. Springer International: Cham, Switzerland, 2015.
-
- Fernández E, Ed. Advances in Organoboron Chemistry towards Organic Synthesis. Thieme: Stuttgart, Germany, 2020.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

