NRX-0492 degrades wild-type and C481 mutant BTK and demonstrates in vivo activity in CLL patient-derived xenografts
- PMID: 36375120
- PMCID: PMC10163313
- DOI: 10.1182/blood.2022016934
NRX-0492 degrades wild-type and C481 mutant BTK and demonstrates in vivo activity in CLL patient-derived xenografts
Abstract
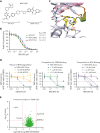
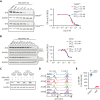
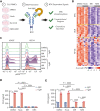
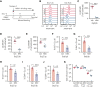
Bruton tyrosine kinase (BTK) is essential for B-cell receptor (BCR) signaling, a driver of chronic lymphocytic leukemia (CLL). Covalent inhibitors bind C481 in the active site of BTK and have become a preferred CLL therapy. Disease progression on covalent BTK inhibitors is commonly associated with C481 mutations. Here, we investigated a targeted protein degrader, NRX-0492, that links a noncovalent BTK-binding domain to cereblon, an adaptor protein of the E3 ubiquitin ligase complex. NRX-0492 selectively catalyzes ubiquitylation and proteasomal degradation of BTK. In primary CLL cells, NRX-0492 induced rapid and sustained degradation of both wild-type and C481 mutant BTK at half maximal degradation concentration (DC50) of ≤0.2 nM and DC90 of ≤0.5 nM, respectively. Sustained degrader activity was maintained for at least 24 hours after washout and was equally observed in high-risk (deletion 17p) and standard-risk (deletion 13q only) CLL subtypes. In in vitro testing against treatment-naïve CLL samples, NRX-0492 was as effective as ibrutinib at inhibiting BCR-mediated signaling, transcriptional programs, and chemokine secretion. In patient-derived xenografts, orally administered NRX-0492 induced BTK degradation and inhibited activation and proliferation of CLL cells in blood and spleen and remained efficacious against primary C481S mutant CLL cells collected from a patient progressing on ibrutinib. Oral bioavailability, >90% degradation of BTK at subnanomolar concentrations, and sustained pharmacodynamic effects after drug clearance make this class of targeted protein degraders uniquely suitable for clinical translation, in particular as a strategy to overcome BTK inhibitor resistance. Clinical studies testing this approach have been initiated (NCT04830137, NCT05131022).
Conflict of interest statement
Conflict-of-interest disclosure: A.K., J.M., A.T.-M., E.A., Y.S.T., H.L., S.G., M.C.C., S.Y., D.W.R., and M.N. were or currently are full-time employees and shareholders of Nurix Therapeutics. A.W. received research funding from Pharmacyclics, Acerta Pharma, Merck, Verastem, Genmab, and Nurix Therapeutics. C.S. received research funding from Genmab. The remaining authors declare no competing financial interests.
Figures




Comment in
-
The cat-and-mouse game of BTK inhibition.Blood. 2023 Mar 30;141(13):1502-1503. doi: 10.1182/blood.2022018936. Blood. 2023. PMID: 36995705 No abstract available.
References
-
- Burger JA. Treatment of chronic lymphocytic leukemia. N Engl J Med. 2020;383(5):460–473. - PubMed
-
- Stevenson FK, Krysov S, Davies AJ, Steele AJ, Packham G. B-cell receptor signaling in chronic lymphocytic leukemia. Blood. 2011;118(16):4313–4320. - PubMed
-
- Treon SP, Tripsas CK, Meid K, et al. IBR in previously treated Waldenstrom's macroglobulinemia. N Engl J Med. 2015;372(15):1430–1440. - PubMed
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

